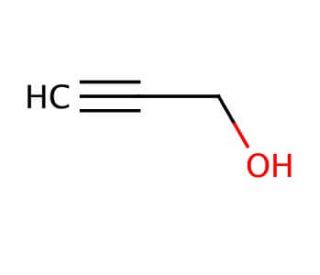
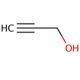
Propargyl alcohol (CAS 107-19-7)
QUICK LINKS
Propargyl alcohol, known by various names such as propynol, prop-2-yn-1-ol, or 2-propyn-1-ol, is an organic compound that finds wide-ranging applications in the scientific and industrial domains. It is a colorless and volatile liquid characterized by a sweet and pungent odor. As a three-carbon alkyl alcohol, its molecular formula is C3H4O. Propargyl alcohol serves as a reagent in organic synthesis and as a solvent for diverse applications. It functions as a catalyst in certain reactions and finds utility as a fuel additive. The applications of propargyl alcohol extend to numerous scientific domains. It plays a vital role in the synthesis of polymers such as poly(ethylene oxide) and poly(propylene oxide). These polymers can possess a wide range of functional groups, such as esters, amides, and amines. Due to its alkylating properties, propargyl alcohol readily reacts with nucleophiles like amines and thiols. Notably, it acts as an oxidizing agent capable of converting primary alcohols to aldehydes and secondary alcohols to ketones. It demonstrates the ability to oxidize sulfides into sulfoxides.
Propargyl alcohol (CAS 107-19-7) References
- Cytochrome P450 2E1 metabolically activates propargyl alcohol: propiolaldehyde-induced hepatocyte cytotoxicity. | Moridani, MY., et al. 2001. Chem Biol Interact. 130-132: 931-42. PMID: 11306107
- A mechanism for the formation of bis-glutathione conjugates of propargyl alcohol. | Banijamali, AR., et al. 2003. Pest Manag Sci. 59: 331-8. PMID: 12639051
- Localization of a substrate binding site on the FeMo-cofactor in nitrogenase: trapping propargyl alcohol with an alpha-70-substituted MoFe protein. | Benton, PM., et al. 2003. Biochemistry. 42: 9102-9. PMID: 12885243
- Toxicology and carcinogenesis studies of propargyl alcohol (CAS No. 107-19-7) in F344/N rats and B6C3F1 mice (inhalation studies). | ,. 2008. Natl Toxicol Program Tech Rep Ser. 1-172. PMID: 18974778
- Tandem reaction of propargyl alcohol and N-sulfonylhydrazone: synthesis of dihydropyrazole and its utility in the preparation of 3,3-diarylacrylonitrile. | Zhu, Y., et al. 2011. Org Lett. 13: 3553-5. PMID: 21661752
- Evaluation of propargyl alcohol toxicity and carcinogenicity in F344/N rats and B6C3F1/N mice following whole-body inhalation exposure. | Thakur, SA., et al. 2013. Toxicology. 314: 100-11. PMID: 24035744
- Thermal decomposition of propargyl alcohol: single pulse shock tube experimental and ab initio theoretical study. | Sharath, N., et al. 2014. J Phys Chem A. 118: 5927-38. PMID: 25036110
- Theoretical Insights into the Catalytic Mechanism of N-Heterocyclic Olefins in Carboxylative Cyclization of Propargyl Alcohol with CO2. | Li, W., et al. 2016. J Org Chem. 81: 5303-13. PMID: 27266858
- Reactions of 1,2-diaza-1,3-butadienes with propargyl alcohol as an approach to novel bi-heterocyclic systems. | De Crescentini, L., et al. 2016. Org Biomol Chem. 14: 8674-8678. PMID: 27714189
- Iron-Catalyzed Dehydrative Alkylation of Propargyl Alcohol with Alkyl Peroxides To Form Substituted 1,3-Enynes. | Ye, C., et al. 2018. Org Lett. 20: 3202-3205. PMID: 29786445
- Silver(i)/base-promoted propargyl alcohol-controlled regio- or stereoselective synthesis of furan-3-carboxamides and (Z)-enaminones. | Sultana, S., et al. 2018. Org Biomol Chem. 16: 6749-6759. PMID: 30187059
- Highly Z-selective synthesis of 1,3-oxathiol-2-ylidenes and 4-methylene-oxazolidine-2-thiones via atom-specific 5-exo-dig cyclization of propargyl alcohol with isothiocyanate. | Antony Savarimuthu, S., et al. 2020. Org Biomol Chem. 18: 3552-3562. PMID: 32342068
- Structure and Internal Motions of a Multifunctional Alcohol-Water Complex: Rotational Spectroscopy of the Propargyl Alcohol···H2O Dimer. | Gnanasekar, SP. and Arunan, E. 2021. J Phys Chem A. 125: 7138-7150. PMID: 34378937
- Experimental and Theoretical DFT Investigations in the [2,3]-Wittig-Type Rearrangement of Propargyl/Allyl-Oxy-Pyrazolones. | Crescentini, L., et al. 2021. Molecules. 26: PMID: 34770965
- Cu-catalyzed, Mn-mediated propargylation and allenylation of aldehydes with propargyl bromides. | Zhang, R., et al. 2022. BMC Chem. 16: 14. PMID: 35303949
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Propargyl alcohol, 100 ml | sc-224210 | 100 ml | $27.00 | |||
Propargyl alcohol, 500 ml | sc-224210A | 500 ml | $37.00 |
