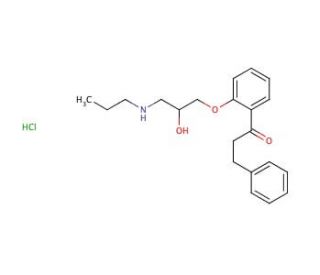
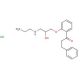
Propafenone Hydrochloride (CAS 34183-22-7)
QUICK LINKS
Propafenone Hydrochloride acts primarily by inhibiting sodium channel proteins, which plays a role in the modulation of electrical signals in cardiac cells, thus affecting the rhythmic contractions of the heart muscle. Beyond its action on sodium channels, it also exhibits inhibitory effects on adrenergic receptors (AR), adding an additional layer to its mechanism of action by influencing the sympathetic nervous system′s control over heart rate and myocardial contractility. This multifaceted mechanism positions Propafenone Hydrochloride as a significant compound in cardiovascular research, particularly in studies aimed at understanding arrhythmias and developing strategies for modulating cardiac electrical activity. Its dual action provides a unique tool for researchers to dissect the complex interplay between ion channels and adrenergic signaling in the heart, making it an invaluable compound in the toolkit for cardiovascular physiology and pharmacology studies.
Propafenone Hydrochloride (CAS 34183-22-7) References
- Efficacy of propafenone hydrochloride in preventing postoperative atrial fibrillation after coronary artery bypass grafting. | Ito, N., et al. 2010. Heart Surg Forum. 13: E223-7. PMID: 20719723
- Host-guest inclusion complex of propafenone hydrochloride with α- and β-cyclodextrins: spectral and molecular modeling studies. | Siva, S., et al. 2013. Spectrochim Acta A Mol Biomol Spectrosc. 115: 559-67. PMID: 23872014
- Attenuated Structural Transformation of Aconitine during Sand Frying Process and Antiarrhythmic Effect of Its Converted Products. | Wang, YJ., et al. 2021. Evid Based Complement Alternat Med. 2021: 7243052. PMID: 34733344
- Attenuated Structural Transformation of Indaconitine during Sand Frying Process and Anti-Arrhythmic Effects of Its Transformed Products. | Wang, Y., et al. 2022. Evid Based Complement Alternat Med. 2022: 8606459. PMID: 35222676
- Investigational New Drug Enabling Nonclinical Safety Pharmacology Assessment of the Iminosugar UV-4, a Broad-Spectrum Host-Targeted Antiviral Agent. | Shearer, J., et al. 2022. Int J Toxicol. 41: 201-211. PMID: 35227115
- Thermodynamic Correlation between Liquid-Liquid Phase Separation and Crystalline Solubility of Drug-Like Molecules. | Uekusa, T., et al. 2022. Pharmaceutics. 14: PMID: 36559054
- Prediction of Liquid-Liquid Phase Separation at the Dissolving Drug Salt Particle Surface. | Uekusa, T. and Sugano, K. 2023. Mol Pharm. 20: 3140-3149. PMID: 37183369
- Beta-blocking and electrophysiological effects of propafenone in volunteers. | Müller-Peltzer, H., et al. 1983. Eur J Clin Pharmacol. 25: 831-3. PMID: 6662183
- Effects of propafenone on TEA-induced action potentials in vascular smooth muscle of canine coronary arteries. | Harder, DR. and Belardinelli, L. 1980. Experientia. 36: 1082-3. PMID: 7418845
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Propafenone Hydrochloride, 1 g | sc-204863 | 1 g | $21.00 | |||
Propafenone Hydrochloride, 5 g | sc-204863A | 5 g | $66.00 | |||
Propafenone Hydrochloride, 25 g | sc-204863B | 25 g | $198.00 | |||
Propafenone Hydrochloride, 100 g | sc-204863C | 100 g | $501.00 |
