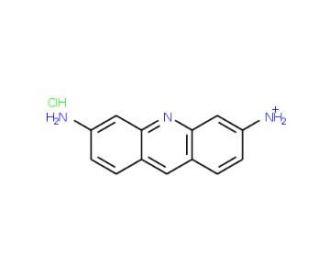
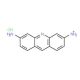
Proflavine Hydrochloride (CAS 952-23-8)
QUICK LINKS
Proflavine Hydrochloride (PFH) is a versatile synthetic compound widely utilized in scientific domains for its antiseptic and antibacterial properties. With its water-soluble, colorless, crystalline form this compound, also known as 1,4-diamino-9,10-anthracenedione hydrochloride, finds extensive applications in laboratory experiments and the synthesis of various compounds.n scientific research, Proflavine Hydrochloride serves as a valuable reagent for studying the effects of antiseptics and antibacterial agents. It is employed to investigate the impact of antibiotics on bacteria, as well as the effects of antiseptics on tissue cultures, viruses, fungi, and other microorganisms.The mode of action of Proflavine Hydrochloride involves the inhibition of bacterial, fungal, and other microorganism growth. This is accomplished by disrupting the cell wall of the microorganisms, impeding their ability to absorb nutrients and reproduce. This mechanism of action shares similarities with other antiseptics and antibacterial agents.
Proflavine Hydrochloride (CAS 952-23-8) References
- Intercalation of proflavine and a platinum derivative of proflavine into double-helical Poly(A). | Ciatto, C., et al. 1999. Biophys J. 77: 2717-24. PMID: 10545371
- Supramolecular solubilisation of hydrophilic dyes by using individual dendritic branches. | Dykes, GM., et al. 2001. Chemistry. 7: 4730-9. PMID: 11757666
- A SINGLE-GENE MUTATION OF CHLAMYDOMONAS REINHARDII AFFECTING MOTILITY: A GENETIC AND ELECTRON MICROSCOPE STUDY. | RANDALL, J., et al. 1964. Nature. 203: 912-4. PMID: 14203504
- UPTAKE OF DYES AND DRUGS BY LIVING CELLS IN CULTURE. | ALLISON, AC. and YOUNG, MR. 1964. Life Sci (1962). 3: 1407-14. PMID: 14248630
- Change of the binding mode of the DNA/proflavine system induced by ethanol. | García, B., et al. 2010. J Phys Chem B. 114: 8555-64. PMID: 20524626
- pH sensitive coiled coils: a strategy for enhanced liposomal drug delivery. | Reja, RM., et al. 2016. Nanoscale. 8: 5139-45. PMID: 26876788
- Dyes in aquaculture and reference points for action. | ,., et al. 2017. EFSA J. 15: e04920. PMID: 32625579
- Excited-State Dynamics of Proflavine after Intercalation into DNA Duplex. | Zhou, J., et al. 2022. Molecules. 27: PMID: 36500248
- Plasmid-mediated resistance to antimicrobial agents among listeriae. | Lemaître, JP., et al. 1998. J Food Prot. 61: 1459-64. PMID: 9829185
- P-type delayed fluorescence from ionic species and aromatic hydrocarbons | C.A. Parker, C.G. Hatchard, Thelma A. Joyce. 1964. Journal of Molecular Spectroscopy. 14: 311-319.
- Concentration quenching of proflavine hydrochloride in dry films of sodium deoxyribonucleate and poly(vinyl alcohol) | G. Strauss, S. B. Broyde, and T. Kurucsev, et al. 1971. J. Phys. Chem. 75: 2727–2733.
- Mono- and bisdiazotization of proflavine | William Firth III and Lerena W. Yielding, et al. 1982. J. Org. Chem. 47: 3002–3004.
- Study on dynamical behaviour of semi-stiff chain polymers. Fluorescence depolarization of proflavine aromatic polyimides | Hideharu Ushiki 1, Mariko Ozu 2. 1986. European Polymer Journal. 22: 835-839.
- Thermodynamic characterization of proflavine–DNA binding through microcalorimetric studies | Anirban Basu, Gopinatha Suresh Kumar. 2015. The Journal of Chemical Thermodynamics. 87: 1-7.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Proflavine Hydrochloride, 10 g | sc-296137 | 10 g | $93.00 | |||
Proflavine Hydrochloride, 25 g | sc-296137A | 25 g | $199.00 |
