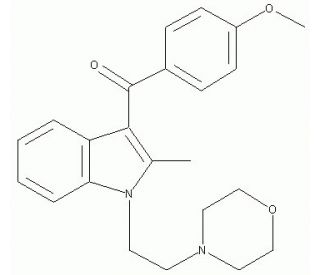
Pravadoline (CAS 92623-83-1)
QUICK LINKS
Pravadoline is a cannabimimetic aminoalkylindole agonist of the cannabinoid receptors and an inhibitor of Cox-1 and Cox-2, demonstrating powerful analgesic effects through the combination of these actions. Pravadoline was developed for Cox inhibition activity and was found to produce analgesic effects at concentrations much lower than needed for Cox inhibition, leading to the observation of Pravadoline-induced inhibition of neuronally mediated smooth muscle contractions distinct from the activity of opioid analgesics. The cannabimimetic activity of Pravadoline was identified in comparison with the binding of CP-55940 (sc-200359), and derivatives of the aminoalkylindole have been subsequently explored towards generating specific cannabinoid receptor agonists.
Pravadoline (CAS 92623-83-1) References
- Aminoalkylindole analogs: cannabimimetic activity of a class of compounds structurally distinct from delta 9-tetrahydrocannabinol. | Compton, DR., et al. 1992. J Pharmacol Exp Ther. 263: 1118-26. PMID: 1335057
- Conformationally restrained analogues of pravadoline: nanomolar potent, enantioselective, (aminoalkyl)indole agonists of the cannabinoid receptor. | D'Ambra, TE., et al. 1992. J Med Chem. 35: 124-35. PMID: 1732519
- [Thiophene as a structural element of physiologically active compounds. 19. The synthesis of substituted (6H-thieno[2,3-b]pyrrol-4-yl)phenylmethanones]. | Binder, D., et al. 1991. Arch Pharm (Weinheim). 324: 219-21. PMID: 1863200
- Antinociceptive (aminoalkyl)indoles. | Bell, MR., et al. 1991. J Med Chem. 34: 1099-110. PMID: 1900533
- Pravadoline: profile in isolated tissue preparations. | Ward, SJ., et al. 1990. J Pharmacol Exp Ther. 255: 1230-9. PMID: 2175798
- Pharmacology of pravadoline: a new analgesic agent. | Haubrich, DR., et al. 1990. J Pharmacol Exp Ther. 255: 511-22. PMID: 2243340
- Combined antiproliferative effects of the aminoalkylindole WIN55,212-2 and radiation in breast cancer cells. | Emery, SM., et al. 2014. J Pharmacol Exp Ther. 348: 293-302. PMID: 24259678
- Pravadoline and aminoalkylindole (AAI) analogues: actions which suggest a receptor interaction. | Ward, SJ., et al. 1989. Br J Pharmacol. 98 Suppl: 831P. PMID: 2611534
- Water-Soluble Hypervalent Iodine(III) Having an I-N Bond. A Reagent for the Synthesis of Indoles. | Xia, HD., et al. 2018. Org Lett. 20: 4052-4056. PMID: 29911872
- The Spicy Story of Cannabimimetic Indoles. | Howlett, AC., et al. 2021. Molecules. 26: PMID: 34684770
- Ni-Catalyzed Reductive Coupling of Alkynes and Amides to Access Multi-Functionalized Indoles. | Min, KH., et al. 2022. Org Lett. 24: 989-994. PMID: 35050641
- (Aminoalkyl)indole isothiocyanates as potential electrophilic affinity ligands for the brain cannabinoid receptor. | Yamada, K., et al. 1996. J Med Chem. 39: 1967-74. PMID: 8642555
- The bioactive conformation of aminoalkylindoles at the cannabinoid CB1 and CB2 receptors: insights gained from (E)- and (Z)-naphthylidene indenes. | Reggio, PH., et al. 1998. J Med Chem. 41: 5177-87. PMID: 9857088
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pravadoline, 5 mg | sc-200369 | 5 mg | $49.00 | |||
Pravadoline, 25 mg | sc-200369A | 25 mg | $261.00 |
