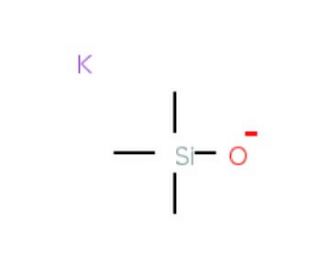
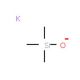
Potassium trimethylsilanolate (CAS 10519-96-7)
QUICK LINKS
Potassium trimethylsilanolate (KTMOS) is an organosilicon compound that finds extensive use in diverse scientific research applications. Potassium trimethylsilanolate exists as a white, crystalline powder that is insoluble in water and has a melting point around 150°C. Its synthesis involves the reaction of trimethylsilanol and potassium hydroxide. Potassium trimethylsilanolate serves as a versatile compound with a unique set of properties, rendering it valuable for various applications. It has been employed as a catalyst in organic reactions, a reagent in organic compound synthesis, and a stabilizer for enzymes. The distinguishing features of potassium trimethylsilanolate contribute to its versatility. It possesses a low melting point, is insoluble in water, and exhibits amphoteric behavior, allowing it to function as either an acid or a base depending on the pH of the solution. Moreover, as an electron-rich compound, Potassium trimethylsilanolate can act as a Lewis acid or a Lewis base. These attributes make potassium trimethylsilanolate useful in a variety of applications across scientific research.
Potassium trimethylsilanolate (CAS 10519-96-7) References
- Potassium trimethylsilanolate induced cleavage of 1,3-oxazolidin-2- and 5-ones, and application to the synthesis of (R)-salmeterol. | Coe, DM., et al. 2003. Org Biomol Chem. 1: 1106-11. PMID: 12926383
- Cross-coupling of alkynylsilanols with aryl halides promoted by potassium trimethylsilanolate. | Denmark, SE. and Tymonko, SA. 2003. J Org Chem. 68: 9151-4. PMID: 14604401
- Validation of the existence of tetrameric species of potassium trimethylsilanolate in the gas phase with a theoretical cluster model: role of the counterion as charge localizer in the structure. | Montejo, M., et al. 2007. J Phys Chem A. 111: 2629-33. PMID: 17388356
- Cationic and anionic surface binding sites on nanocrystalline zinc oxide: surface influence on photoluminescence and photocatalysis. | Bohle, DS. and Spina, CJ. 2009. J Am Chem Soc. 131: 4397-404. PMID: 19265384
- Anhydrous, Homogeneous, Suzuki-Miyaura Cross-Coupling of Boronic Esters using Potassium Trimethylsilanolate. | Delaney, CP., et al. 2020. Organic Synth. 97: 245-261. PMID: 33456091
- Potassium Trimethylsilanolate Enables Rapid, Homogeneous Suzuki-Miyaura Cross-Coupling of Boronic Esters. | Delaney, CP., et al. 2020. ACS Catal. 10: 73-80. PMID: 33585070
- Heteroaryl-Heteroaryl, Suzuki-Miyaura, Anhydrous Cross-Coupling Reactions Enabled by Trimethyl Borate. | Kassel, VM., et al. 2021. J Am Chem Soc. 143: 13845-13853. PMID: 34415757
- Potassium Trimethylsilanolate-Promoted, Anhydrous Suzuki-Miyaura Cross-Coupling Reaction Proceeds via the 'Boronate Mechanism': Evidence for the Alternative Fork in the Trail. | Delaney, CP., et al. 2022. J Am Chem Soc. 144: 4345-4364. PMID: 35230833
- Efficient synthesis of pentasubstituted pyrroles via intramolecular C-arylation. | Lemrová, B., et al. 2022. Org Biomol Chem. 20: 3811-3816. PMID: 35467690
- Suzuki C-C Coupling in Paper Spray Ionization: Microsynthesis of Biaryls and High-Sensitivity MS Detection of Aryl Bromides. | Lin, Q., et al. 2022. J Am Soc Mass Spectrom. 33: 1921-1935. PMID: 36074999
- Synthesis of Benzhydryl-Substituted Amines by Silanolate-Mediated Aldimine Arylation with Functionalized Aryl Nucleophiles Released from Diazene-Based Reagents. | Rahman, AJM., et al. 2022. Org Lett. 24: 9118-9122. PMID: 36469587
- Synthesis of Polysubstituted Pyridines and Pyrazines via Truce-Smiles Rearrangement of Amino Acid-Based 4-Nitrobenzenesulfonamides. | Tkadlecová, M., et al. 2023. J Org Chem. 88: 3228-3237. PMID: 36797215
- Automated carboxy-terminal sequence analysis of polypeptides containing C-terminal proline. | Bailey, JM., et al. 1995. Anal Biochem. 224: 588-96. PMID: 7733462
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Potassium trimethylsilanolate, 25 g | sc-236458 | 25 g | $70.00 |
