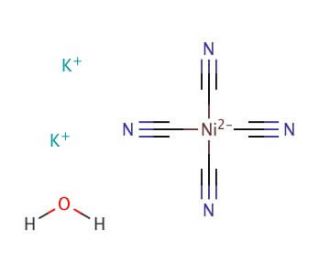
Molecular structure of Potassium tetracyanonickelate(II), CAS Number: 14220-17-8
Potassium tetracyanonickelate(II) (CAS 14220-17-8)
Alternate Names:
Dipotassium nickel(2+) tetracyanide
Application:
Potassium tetracyanonickelate(II) is a useful biochemical for proteomics research
CAS Number:
14220-17-8
Molecular Weight:
240.96
Molecular Formula:
K2Ni(CN)4
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Potassium tetracyanonickelate(II) hydrate acts as a catalyst in various chemical reactions, particularly in organic synthesis. It facilitates the formation of carbon-carbon bonds and is involved in the production of intermediates and fine chemicals. The compound′s mechanism of action involves its ability to coordinate with other molecules, allowing for the activation of specific chemical bonds and the promotion of desired reactions. This coordination complex plays a role in electron transfer processes, enabling the transformation of substrates into products. Its function as a catalyst in various reactions may be useful in the development of new compounds and materials.
Potassium tetracyanonickelate(II) (CAS 14220-17-8) References
- Electrochemical and XPS characterization of composite modified electrodes obtained by nickel deposition on noble metals. | Casella, IG. and Gatta, M. 2000. Anal Chem. 72: 2969-75. PMID: 10905336
- Substrate-regulated cyanide hydratase (chy) gene expression in Fusarium solani: the potential of a transcription-based assay for monitoring the biotransformation of cyanide complexes. | Barclay, M., et al. 2002. Environ Microbiol. 4: 183-9. PMID: 12000318
- The impact of bioaugmentation on metal cyanide degradation and soil bacteria community structure. | Baxter, J. and Cummings, SP. 2006. Biodegradation. 17: 207-17. PMID: 16715400
- Synthesis and characterization of lithium oxonitrate (LiNO). | Switzer, CH., et al. 2013. J Inorg Biochem. 118: 128-33. PMID: 23107606
- Chemically Binding Scaffolded Anodes with 3D Graphene Architectures Realizing Fast and Stable Lithium Storage. | Wu, P., et al. 2019. Research (Wash D C). 2019: 8393085. PMID: 31549090
- Hierarchical Metal-[Carbon Nitride Shell/Carbon Core] Electrocatalysts: A Promising New General Approach to Tackle the ORR Bottleneck in Low-Temperature Fuel Cells. | Di Noto, V., et al. 2022. ACS Catal. 12: 12291-12301. PMID: 36249870
- Synthesis of Prussian Blue Analogue and Its Catalytic Activity toward Reduction of Environmentally Toxic Nitroaromatic Pollutants. | El Mously, DA., et al. 2022. ACS Omega. 7: 43139-43146. PMID: 36467928
- Facile synthesis of porous transition metal hydroxides from a poly(4-vinyl pyridine) film by controlling pH. | Ha, G., et al. 2023. Nanoscale Adv. 5: 2565-2572. PMID: 37143805
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Potassium tetracyanonickelate(II), 100 g | sc-228977 | 100 g | $145.00 |
