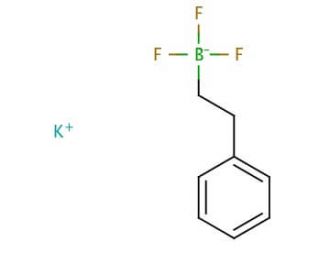
Potassium phenethyltrifluoroborate (CAS 329976-74-1)
QUICK LINKS
Potassium phenethyltrifluoroborate is extensively studied in organic chemistry, particularly for its applications in Suzuki-Miyaura cross-coupling reactions. This compound is utilized for its ability to act as a stable and efficient source of the phenethyl group in the formation of carbon-carbon bonds. Research on Potassium phenethyltrifluoroborate includes investigations into its reactivity and efficiency in various organic synthesis conditions, focusing on improving yield and selectivity of desired products. Studies also explore the solubility and stability of Potassium phenethyltrifluoroborate in different solvents, which is for its practical use in synthetic chemistry. Moreover, the environmental impact of using Potassium phenethyltrifluoroborate is assessed to ensure that its application is sustainable and safe within industrial and laboratory applications.
Potassium phenethyltrifluoroborate (CAS 329976-74-1) References
- The synthesis of 5-substituted ring E analogs of methyllycaconitine via the Suzuki-Miyaura cross-coupling reaction. | Huang, J., et al. 2008. Bioorg Med Chem. 16: 3816-24. PMID: 18272373
- Suzuki-Miyaura cross-coupling reactions of primary alkyltrifluoroborates with aryl chlorides. | Dreher, SD., et al. 2009. J Org Chem. 74: 3626-31. PMID: 19271726
- Oxidation of organotrifluoroborates via oxone. | Molander, GA. and Cavalcanti, LN. 2011. J Org Chem. 76: 623-30. PMID: 21192650
- Copper-catalyzed oxidative Heck reactions between alkyltrifluoroborates and vinyl arenes. | Liwosz, TW. and Chemler, SR. 2013. Org Lett. 15: 3034-7. PMID: 23734764
- Visible light-promoted alkylation of imines using potassium organotrifluoroborates. | Plasko, DP., et al. 2018. Photochem Photobiol Sci. 17: 534-538. PMID: 29722411
- Electrochemical cobalt-catalyzed C-H or N-H oxidation: a facile route to synthesis of substituted oxindoles. | Yu, Y., et al. 2018. Org Biomol Chem. 16: 8917-8921. PMID: 30427366
- Recent Advances in Photoredox-Mediated Radical Conjugate Addition Reactions: An Expanding Toolkit for the Giese Reaction. | Gant Kanegusuku, AL. and Roizen, JL. 2021. Angew Chem Int Ed Engl. 60: 21116-21149. PMID: 33629454
- Access to chiral β-sulfonyl carbonyl compounds via photoinduced organocatalytic asymmetric radical sulfonylation with sulfur dioxide. | He, FS., et al. 2022. Chem Sci. 13: 8834-8839. PMID: 35975150
- Photocatalytic Sulfonyl Fluorination of Alkyl Organoboron Substrates. | Vincent, CA., et al. 2023. ACS Catal. 13: 3668-3675. PMID: 37124721
- Redox-economical radical generation from organoborates and carboxylic acids by organic photoredox catalysis | Tatsuya Chinzei, Kazuki Miyazawa, Yusuke Yasu, Takashi Koike* and Munetaka Akita*. 2015. RSC Adv. 5: 21297-21300.
- Electrochemical Oxidation of Organotrifluoroborate Compounds | Junji Suzuki, Masahiro Tanigawa, Prof. Dr. Shinsuke Inagi, Prof. Dr. Toshio Fuchigami. 2016. ChemElectroChem. 3: 2078-2083.
- Electrochemical properties and reactions of organoboron compounds | Shinsuke Inagi 1, Toshio Fuchigami 2. 2017. Current Opinion in Electrochemistry. 2: 32-37.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Potassium phenethyltrifluoroborate, 1 g | sc-236452 | 1 g | $133.00 |
