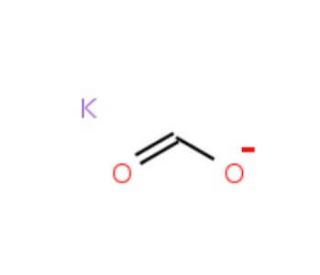
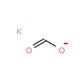
Potassium formate solution (CAS 590-29-4)
QUICK LINKS
Potassium formate solution, a water-soluble potassium salt of formic acid, is primarily utilized in scientific research for its properties as a buffering agent and a source of formate ions. Its ability to generate a stable pH environment makes it valuable in biochemical studies, particularly in enzyme kinetics and other reactions sensitive to pH fluctuations. Researchers leverage potassium formate to investigate enzyme mechanisms where formate acts as either a substrate or a product, enabling detailed studies on enzyme activity and substrate specificity. Additionally, its high ionic strength and minimal interference in optical clarity make it suitable for use in spectroscopic studies, including nuclear magnetic resonance (NMR) and mass spectrometry, to study protein folding and molecular interactions. In molecular biology, potassium formate is utilized in the preparation of nucleic acid samples for PCR amplification, where it enhances the efficiency of the process without degrading the nucleic acids. Its role extends to biotechnological applications, such as fermentation processes, where it serves as an ionic mediator and osmoprotectant, contributing to the stability and viability of microbial cultures under stress conditions. This versatility underscores potassium formate′s importance in advancing the understanding of complex biological and chemical systems.
Potassium formate solution (CAS 590-29-4) References
- Bacterial formate hydrogenlyase complex. | McDowall, JS., et al. 2014. Proc Natl Acad Sci U S A. 111: E3948-56. PMID: 25157147
- Investigations by Protein Film Electrochemistry of Alternative Reactions of Nickel-Containing Carbon Monoxide Dehydrogenase. | Wang, VC., et al. 2015. J Phys Chem B. 119: 13690-7. PMID: 26176986
- Occurrence of microplastics in edible aquatic insect Pantala sp. (Odonata: Libellulidae) from rice fields. | Maneechan, W. and Prommi, TO. 2022. PeerJ. 10: e12902. PMID: 35186487
- Nutritional Compositions of Aquatic Insects Living in Rice Fields, with a Particular Focus on Odonate Larvae. | Maneechan, W., et al. 2022. Insects. 13: PMID: 36555041
- Resolution of 1, 2-diols by enzyme-catalyzed oxidation with anodic, mediated cofactor regeneration in the extractive membrane reactor: gaining insight by adaptive simulation. | Degenring, Daniela, et al. 2004. Organic process research & development. 8.2: 213-218.
- A method to create a universal calibration dataset for Raman reconstruction based on wiener estimation. | Chen, Shuo, et al. 2015. IEEE Journal of selected topics in quantum electronics. 22.3: 164-170.
- Manipulation of gas-liquid-liquid systems in continuous flow microreactors for efficient reaction processes. | Liu, Yanyan, et al. 2020. Journal of Flow Chemistry. 10.1: 103-121.
- Continuous solid particle flow in microreactors for efficient chemical conversion. | Zong, Jie and Jun Yue. 2022. ndustrial & Engineering Chemistry Research. 61.19: 6269-6291.
- Effects of Salty Additives in Drilling Fluids on Hydrate Formation Dynamics and Phase Equilibrium. | Song, Lifang, et al. 2023. Energy & Fuels. 37.13: 9089-9101.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Potassium formate solution, 100 ml | sc-301590 | 100 ml | $59.00 | |||
Potassium formate solution, 500 ml | sc-301590A | 500 ml | $213.00 |
