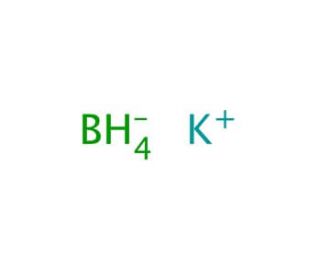

Molecular structure of Potassium borohydride, CAS Number: 13762-51-1
Potassium borohydride (CAS 13762-51-1)
See product citations (1)
Alternate Names:
Potassium tetrahydroborate
Application:
Potassium borohydride is a chemical used in synthesis as a selective reducing agent
CAS Number:
13762-51-1
Molecular Weight:
53.94
Molecular Formula:
K•BH4
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
Available in US only.
Available in US only.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Potassium borohydride a chemical used in synthesis as a selective reducing agent. It is very selective and as such will only reduce ketones or aldehydes to their corresponding alcohols. It is used to synthesize chloramphenicol, vitamin A, thiopenicol, atropine, and scopolamine. It differs from other borohydrides as it is insoluble in ethanol, ether, and THF.
Potassium borohydride (CAS 13762-51-1) References
- Reduction of nitroarenes to azoxybenzenes by potassium borohydride in water. | Liu, Y., et al. 2011. Molecules. 16: 3563-8. PMID: 21527883
- Asymmetric 1,2-reduction of enones with potassium borohydride catalyzed by chiral N,N'-dioxide-scandium(III) complexes. | He, P., et al. 2012. Org Lett. 14: 5134-7. PMID: 23013322
- The effect of copper on iron reduction and its application to the determination of total iron content in iron and copper ores by potassium dichromate titration. | Hu, H., et al. 2014. Talanta. 125: 425-31. PMID: 24840467
- Eutectic melting of LiBH4-KBH4. | Ley, MB., et al. 2014. Phys Chem Chem Phys. 16: 24194-9. PMID: 25293724
- Catalytic reduction of 2-nitroaniline: a review. | Naseem, K., et al. 2017. Environ Sci Pollut Res Int. 24: 6446-6460. PMID: 28054271
- Fabrication of Silver Decorated Graphene Oxide Composite for Photocatalytic Inactivation of Escherichia coli. | Zhang, H., et al. 2018. J Nanosci Nanotechnol. 18: 2304-2309. PMID: 29442896
- ISOBAM-stabilized Ni2+ colloidal catalysts: high catalytic activities for hydrogen generation from hydrolysis of KBH4. | Pei, Y., et al. 2020. Nanotechnology. 31: 134003. PMID: 31783396
- Heats of Hydrolysis and Formation of Potassium Borohydride. | Johnson, WH., et al. 1961. J Res Natl Bur Stand A Phys Chem. 65A: 97-99. PMID: 32196230
- Safer plant-based nanoparticles for combating antibiotic resistance in bacteria: A comprehensive review on its potential applications, recent advances, and future perspective. | Anand, U., et al. 2022. Sci Total Environ. 821: 153472. PMID: 35093375
- Multifunctional Surface Construction for Long-Term Cycling Stability of Li-Rich Mn-Based Layered Oxide Cathode for Li-Ion Batteries. | Yan, C., et al. 2022. Small. 18: e2107910. PMID: 35768284
- Fe3O4@SiO2-PMA-Cu magnetic nanoparticles as a novel catalyst for green synthesis of β-thiol-1,4-disubstituted-1,2,3-triazoles. | Eisavi, R. and Ahmadi, F. 2022. Sci Rep. 12: 11939. PMID: 35831386
- Single-factor analysis of Ni-B-AC-catalyzed β-pinene hydrogenation based on hierarchical analysis. | Deng, Q., et al. 2022. RSC Adv. 12: 28560-28571. PMID: 36320501
- Rhodopsin of the larval mosquito. | Brown, PK. and White, RH. 1972. J Gen Physiol. 59: 401-14. PMID: 5029551
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Potassium borohydride, 5 g | sc-250747 | 5 g | $65.00 | |||
| US: Only available in the US | ||||||
