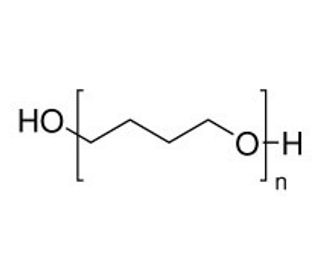
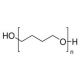
Poly(tetrahydrofuran) (CAS 25190-06-1)
QUICK LINKS
Poly(tetrahydrofuran) (PTHF) is a polyether that can be synthesized through cationic polymerization of tetrahydrofuran in the presence of various initiator systems. Poly(tetrahydrofuran) exhibits the characteristics of a thermoplastic elastomer, offering exceptional chemical resistance, favorable mechanical properties, and excellent thermal stability. It has found utility as a support matrix in cell culture applications, as it can be easily modified to create an optimal environment for cell growth. Composed of elongated chain molecules, Poly(tetrahydrofuran) relies on covalent bonds formed between the oxygen atoms of the THF monomer units to establish cohesion. Additionally, hydrogen bonds formed between the hydrogen atoms of the THF monomer units contribute to the intermolecular strength. These covalent and hydrogen bonds contribute to the thermoplastic nature of the polymer, allowing it to be heated and reshaped into different forms as desired.
Poly(tetrahydrofuran) (CAS 25190-06-1) References
- Molecular recognition in a thermoplastic elastomer. | Koevoets, RA., et al. 2005. J Am Chem Soc. 127: 2999-3003. PMID: 15740137
- Polymer grafting onto starch nanocrystals. | Labet, M., et al. 2007. Biomacromolecules. 8: 2916-27. PMID: 17718501
- Iron-catalyzed ring-closing depolymerization of poly(tetrahydrofuran). | Enthaler, S. and Trautner, A. 2013. ChemSusChem. 6: 1334-6. PMID: 23821541
- Probing Force with Mechanobase-Induced Chemiluminescence. | Clough, JM., et al. 2016. Angew Chem Int Ed Engl. 55: 1445-9. PMID: 26663819
- Cross-linked poly(tetrahydrofuran) as promising sorbent for organic solvent/oil spill. | Yati, I., et al. 2016. J Hazard Mater. 309: 210-8. PMID: 26894295
- An improved fabric-phase sorptive extraction protocol for the determination of seven parabens in human urine by HPLC-DAD. | Rigkos, G., et al. 2021. Biomed Chromatogr. 35: e4974. PMID: 32893361
- Tribological evaluation of a novel hybrid for repair of articular cartilage defects. | Parkes, M., et al. 2021. Mater Sci Eng C Mater Biol Appl. 119: 111495. PMID: 33321596
- Silver- and Zinc-Decorated Polyurethane Ionomers with Tunable Hard/Soft Phase Segregation. | Rapone, I., et al. 2021. Int J Mol Sci. 22: PMID: 34200185
- Metallosupramolecular polymers as precursors for platinum nanocomposites. | Cappelletti, C., et al. 2022. Polym Chem. 13: 1880-1890. PMID: 35432604
- Systematic Synthesis of Block Copolymers Consisting of Topological Amphiphilic Segment Pairs from kyklo- and kentro-Telechelic PEO and Poly(THF). | Hatakeyama, F., et al. 2013. ACS Macro Lett. 2: 427-431. PMID: 35581851
- Designing an 'all-in-one' microextraction capsule device for the liquid chromatographic-fluorescence determination of doxorubicin and its metabolites in rat plasma. | Mamounas, G., et al. 2022. J Chromatogr A. 1680: 463432. PMID: 36041251
- In vivo testing of crosslinked polyethers. I. Tissue reactions and biodegradation. | Pol, BJ., et al. 1996. J Biomed Mater Res. 32: 307-20. PMID: 8897135
- Surface functionalization of cellulose by grafting oligoether chains | J Bras, P Sadocco, MN Belgacem, A Dufresne. 2010. Materials Chemistry and Physics. 120, 2–3: 438-445.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Poly(tetrahydrofuran), 1 L | sc-255437 | 1 L | $240.00 |
