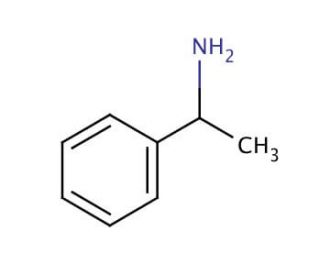
(±)-α-Methylbenzylamine (CAS 618-36-0)
QUICK LINKS
(±)-α-Methylbenzylamine is an organic compound categorized as an alkylamine. Present in minute quantities in certain plants, as well as in some foods, it serves as a precursor for various essential compounds, notably the neurotransmitter dopamine and the hormone epinephrine. Beyond its natural occurrence, (±)-α-Methylbenzylamine finds extensive applications in scientific research. In the realm of scientific investigation, (±)-α-Methylbenzylamine plays a pivotal role in the synthesis of other substances like neurotransmitters and hormones. Furthermore, it serves as a substrate for enzyme reactions, such as facilitating the conversion of tyrosine into dopamine. (±)-α-Methylbenzylamine boasts diverse applications in scientific exploration, showcasing its significance in the synthesis of vital compounds.
(±)-α-Methylbenzylamine (CAS 618-36-0) References
- Kinetic modeling of omega-transamination for enzymatic kinetic resolution of alpha-methylbenzylamine. | Shin, JS. and Kim, BG. 1998. Biotechnol Bioeng. 60: 534-40. PMID: 10099461
- Deracemization of alpha-methylbenzylamine using an enzyme obtained by in vitro evolution. | Alexeeva, M., et al. 2002. Angew Chem Int Ed Engl. 41: 3177-80. PMID: 12207381
- Simultaneous synthesis of enantiomerically pure (R)-1-phenylethanol and (R)-alpha-methylbenzylamine from racemic alpha-methylbenzylamine using omega-transaminase/alcohol dehydrogenase/glucose dehydrogenase coupling reaction. | Yun, H., et al. 2003. Biotechnol Lett. 25: 809-14. PMID: 12882012
- A study of the liquid and solvent properties of optically active and racemic alpha-methylbenzylamine. | Fischer, AT., et al. 2007. J Phys Chem B. 111: 8187-96. PMID: 17583940
- Simple protocols for NMR analysis of the enantiomeric purity of chiral diols. | Kelly, AM., et al. 2008. Nat Protoc. 3: 215-9. PMID: 18274523
- Rapid and sensitive kinetic assay for characterization of omega-transaminases. | Schätzle, S., et al. 2009. Anal Chem. 81: 8244-8. PMID: 19739593
- Three-component chiral derivatizing protocols for NMR spectroscopic enantiodiscrimination of hydroxy acids and primary amines. | Chaudhari, SR. and Suryaprakash, N. 2012. J Org Chem. 77: 648-51. PMID: 22145800
- ω-Transaminase from Ochrobactrum anthropi is devoid of substrate and product inhibitions. | Park, ES. and Shin, JS. 2013. Appl Environ Microbiol. 79: 4141-4. PMID: 23584786
- Natural product derived antiprotozoal agents: synthesis, biological evaluation, and structure-activity relationships of novel chromene and chromane derivatives. | Harel, D., et al. 2013. J Med Chem. 56: 7442-8. PMID: 23968432
- Tailoring D-amino acid oxidase from the pig kidney to R-stereoselective amine oxidase and its use in the deracemization of α-methylbenzylamine. | Yasukawa, K., et al. 2014. Angew Chem Int Ed Engl. 53: 4428-31. PMID: 24644036
- Looking Inside the Intramolecular C-H∙∙∙O Hydrogen Bond in Lactams Derived from α-Methylbenzylamine. | Mejía, S., et al. 2017. Molecules. 22: PMID: 28264508
- Self-Assembly of Peptide Chiral Nanostructures with Sequence-Encoded Enantioseparation Capability. | Fan, Y., et al. 2020. Langmuir. 36: 10361-10370. PMID: 32787008
- Construction and Application of Graphene Oxide-Bovine Serum Albumin Modified Extended Gate Field Effect Transistor Chiral Sensor. | Li, L., et al. 2021. Sensors (Basel). 21: PMID: 34200213
- Studies of monoamine oxidase and semicarbazide-sensitive amine oxidase. II. Inhibition by alpha-methylated substrate-analogue monoamines, alpha-methyltryptamine, alpha-methylbenzylamine and two enantiomers of alpha-methylbenzylamine. | Arai, Y., et al. 1986. Jpn J Pharmacol. 41: 191-7. PMID: 3747266
- Probes of the active site of norepinephrine N-methyltransferase: effect of hydrophobic and hydrophilic interactions on side-chain binding of amphetamine and alpha-methylbenzylamine. | Grunewald, GL., et al. 1982. J Med Chem. 25: 1248-50. PMID: 7143366
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(±)-α-Methylbenzylamine, 25 ml | sc-239163 | 25 ml | $68.00 | |||
(±)-α-Methylbenzylamine, 100 ml | sc-239163A | 100 ml | $89.00 |
