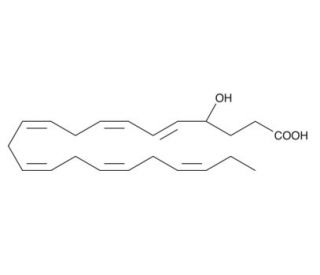
(±)4-HDoHE (CAS 90906-40-4)
See product citations (1)
QUICK LINKS
(±)4-HDoHE, registered under CAS number 90906-40-4, is a hydroxydocosahexaenoic acid derivative, specifically involving the docosahexaenoic acid (DHA), which is an omega-3 fatty acid primarily found in marine oils like fish oil. This compound features a hydroxyl group at the fourth carbon and includes both enantiomers, as indicated by the (±) notation. In scientific research, (±)4-HDoHE is explored for its potential role in modulating cellular signaling pathways related to inflammation and other cellular functions. As a lipid mediator, it is particularly intriguing due to its interaction with various enzymes and receptors that influence cellular signaling networks. These interactions are pivotal in regulating the synthesis and activity of a range of bioactive molecules, including cytokines and eicosanoids, which are crucial in managing the body′s inflammatory responses. Studies focusing on (±)4-HDoHE aim to delineate its effects on the complex pathways of lipid metabolism and signaling. By understanding how this compound influences these pathways, researchers can gain insights into the broader biological roles of omega-3 fatty acids. Such research is vital for advancing our knowledge of cellular mechanisms that control inflammation, cellular protection, and homeostasis, ultimately contributing to our understanding of how dietary factors can influence cellular health and function.
(±)4-HDoHE (CAS 90906-40-4) References
- Human paraoxonases (PON1, PON2, and PON3) are lactonases with overlapping and distinct substrate specificities. | Draganov, DI., et al. 2005. J Lipid Res. 46: 1239-47. PMID: 15772423
- 5-Lipoxygenase metabolite 4-HDHA is a mediator of the antiangiogenic effect of ω-3 polyunsaturated fatty acids. | Sapieha, P., et al. 2011. Sci Transl Med. 3: 69ra12. PMID: 21307302
- Stereochemical analysis of hydroxylated docosahexaenoates produced by human platelets and rat brain homogenate. | Kim, HY., et al. 1990. Prostaglandins. 40: 473-90. PMID: 2147773
- Relationship between the omega-3 index and specialized pro-resolving lipid mediators in patients with peripheral arterial disease taking fish oil supplements. | Schaller, MS., et al. 2017. J Clin Lipidol. 11: 1289-1295. PMID: 28778393
- Oxylipin responses to fasting and insulin infusion in a large mammalian model of fasting-induced insulin resistance, the northern elephant seal. | Wright, DN., et al. 2021. Am J Physiol Regul Integr Comp Physiol. 321: R537-R546. PMID: 34346724
- Dietary Omega-3 Polyunsaturated Fatty Acids and Amelioration of CKD: Possible Cellular Mechanisms. | Yamamoto, T. and Isaka, Y. 2023. Kidney360. 4: 1661-1662. PMID: 38153790
- Oxidation of docosahexaenoic acid by rat liver microsomes. | VanRollins, M., et al. 1984. J Biol Chem. 259: 5776-83. PMID: 6232277
- Autooxidation of docosahexaenoic acid: analysis of ten isomers of hydroxydocosahexaenoate. | VanRollins, M. and Murphy, RC. 1984. J Lipid Res. 25: 507-17. PMID: 6234372
- Docosahexaenoic acid is a strong inhibitor of prostaglandin but not leukotriene biosynthesis. | Corey, EJ., et al. 1983. Proc Natl Acad Sci U S A. 80: 3581-4. PMID: 6304720
- High-performance liquid chromatography-thermospray mass spectrometry of epoxy polyunsaturated fatty acids and epoxyhydroxy polyunsaturated fatty acids from an incubation mixture of rat tissue homogenate. | Yamane, M., et al. 1994. J Chromatogr. 652: 123-36. PMID: 8006098
- Facile preparation and structural determination of monohydroxy derivatives of docosahexaenoic acid (HDoHE) by alpha-tocopherol-directed autoxidation. | Reynaud, D., et al. 1993. Anal Biochem. 214: 165-70. PMID: 8250220
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(±)4-HDoHE, 25 µg | sc-205120 | 25 µg | $60.00 | |||
(±)4-HDoHE, 50 µg | sc-205120A | 50 µg | $154.00 |
