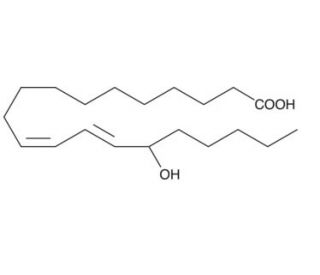
(±)15-HEDE (CAS 77159-57-0)
QUICK LINKS
(±)15-HEDE, bearing the CAS number 77159-57-0, is a racemic mixture of 15-hydroxyeicosa-5(Z),8(Z),11(Z),13(E)-tetraenoic acid enantiomers. This chemical is a derivative of arachidonic acid, produced through enzymatic oxidation involving lipoxygenases. The presence of both enantiomers in the racemic mix allows for the exploration of stereochemical influences on biological activity in research settings. In cellular and biochemical studies, (±)15-HEDE has been instrumental in investigating the regulation of signaling pathways that influence cellular responses to oxidative stress and inflammation. The compound operates by modulating the activities of key signaling proteins and enzymes, impacting processes such as the regulation of gene expression, modulation of receptor activity, and alteration in ion channel function. The specific biochemical roles of (±)15-HEDE include its interaction with lipid signaling pathways, which are crucial for understanding how cells adapt to and communicate under various physiological conditions. Research utilizing this compound often focuses on its ability to influence these pathways, providing insights into the complex network of interactions that govern cellular responses and homeostasis. This detailed understanding aids in elucidating the broader implications of lipid mediators in cellular function and response mechanisms across a variety of biological contexts.
(±)15-HEDE (CAS 77159-57-0) References
- The Nicotiana attenuata GLA1 lipase controls the accumulation of Phytophthora parasitica-induced oxylipins and defensive secondary metabolites. | Schuck, S., et al. 2014. Plant Cell Environ. 37: 1703-15. PMID: 24450863
- A comprehensive quantification method for eicosanoids and related compounds by using liquid chromatography/mass spectrometry with high speed continuous ionization polarity switching. | Yamada, M., et al. 2015. J Chromatogr B Analyt Technol Biomed Life Sci. 995-996: 74-84. PMID: 26046978
- Profiling of serum oxylipins identifies distinct spectrums and potential biomarkers in young people with very early onset gout. | Wang, C., et al. 2023. Rheumatology (Oxford). 62: 1972-1979. PMID: 36111871
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(±)15-HEDE, 25 µg | sc-205021 | 25 µg | $68.00 | |||
(±)15-HEDE, 50 µg | sc-205021A | 50 µg | $129.00 |
