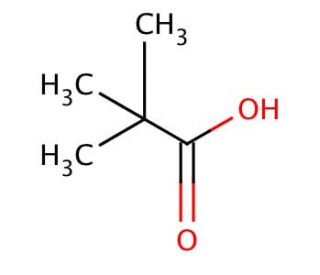
Pivalic acid (CAS 75-98-9)
QUICK LINKS
Pivalic acid functions as a carboxylic acid derivative in experimental applications. It acts as a precursor in the synthesis of various agrochemicals, and other organic compounds. Pivalic acid participates in esterification reactions, where it is used to form pivalate esters, which are important intermediates in organic synthesis. Its mechanism of action involves its ability to donate a proton to other molecules, facilitating the formation of new chemical bonds. Pivalic Acid plays a role in the modification of organic molecules, allowing for the creation of diverse compounds with specific properties. In experimental applications, pivalic acid is utilized as a building block for the preparation of various chemical substances, contributing to the development of new materials and compounds.
Pivalic acid (CAS 75-98-9) References
- Anaerobic mineralization of quaternary carbon atoms: isolation of denitrifying bacteria on pivalic acid (2,2-dimethylpropionic acid). | Probian, C., et al. 2003. Appl Environ Microbiol. 69: 1866-70. PMID: 12620885
- Involvement of recognition and interaction of carnitine transporter in the decrease of L-carnitine concentration induced by pivalic acid and valproic acid. | Okamura, N., et al. 2006. Pharm Res. 23: 1729-35. PMID: 16826461
- Children's toxicology from bench to bed--Liver injury (1): Drug-induced metabolic disturbance--toxicity of 5-FU for pyrimidine metabolic disorders and pivalic acid for carnitine metabolism. | Ito, T. 2009. J Toxicol Sci. 34 Suppl 2: SP217-22. PMID: 19571472
- Pivalic acid acts as a starter unit in a fatty acid and antibiotic biosynthetic pathway in Alicyclobacillus, Rhodococcus and Streptomyces. | Rezanka, T., et al. 2011. Environ Microbiol. 13: 1577-89. PMID: 21450004
- Primary carnitine deficiency and pivalic acid exposure causing encephalopathy and fatal cardiac events. | Rasmussen, J., et al. 2013. J Inherit Metab Dis. 36: 35-41. PMID: 22566287
- Biosynthesis and metabolic pathways of pivalic acid. | Rezanka, T., et al. 2012. Appl Microbiol Biotechnol. 95: 1371-6. PMID: 22790609
- Newborn hypocarnitinemia due to long-term transplacental pivalic acid passage. | Nasu, T., et al. 2014. Pediatr Int. 56: 772-4. PMID: 25335995
- Hypocarnitinemia Observed in an Infant Treated with Short-Term Administration of Antibiotic Containing Pivalic Acid. | Nakazaki, K., et al. 2018. Tohoku J Exp Med. 244: 279-282. PMID: 29628457
- An alternative method for the synthesis of 2'-halogeno-1',2'-unsaturated uridine derivatives through syn-elimination of pivalic acid of 2'-halogeno- 2'-deoxy-1'-pivaloyloxyuracil nucleoside: preparation of its 2'-C-branched nucleosides. | Haraguchi, K., et al. 2020. Nucleosides Nucleotides Nucleic Acids. 39: 426-438. PMID: 31429364
- Microwave and Computational Study of Pivalic Sulfuric Anhydride and the Pivalic Acid Monomer: Mechanistic Insights into the RCOOH + SO3 Reaction. | Love, N., et al. 2022. J Phys Chem A. 126: 6194-6202. PMID: 36067456
- Comparison of ocular disposition of free pivalic acid and pivalic acid esterified in dipivefrin. | Tamaru, RD., et al. 1983. Arch Ophthalmol. 101: 1127-9. PMID: 6347153
- A new esterase for the cleavage of pivalic acid-containing prodrug esters of cephalosporins. | Sauber, K., et al. 1996. Enzyme Microb Technol. 19: 15-9. PMID: 8672280
- Pivalic acid-induced carnitine deficiency and physical exercise in humans. | Abrahamsson, K., et al. 1996. Metabolism. 45: 1501-7. PMID: 8969283
- The use of pivalic acid as a reference substance in measurements of production of volatile fatty acids by rumen micro-organisms in vitro. | Czerkawski, JW. 1976. Br J Nutr. 36: 311-5. PMID: 952843
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pivalic acid, 5 ml | sc-250736 | 5 ml | $20.00 | |||
Pivalic acid, 100 ml | sc-250736A | 100 ml | $27.00 |
