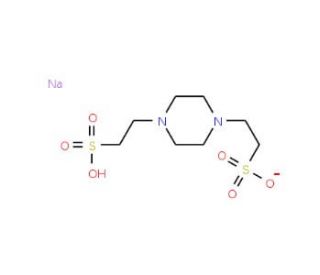
PIPES sodium salt (CAS 10010-67-0)
QUICK LINKS
PIPES sodium salt, a derivative of the parent compound PIPES (Piperazine-N,N′-bis(2-ethanesulfonic acid)), functions as a highly effective biological buffer, commonly utilized in research environments that demand precise pH control. This buffer is particularly valued for its stability and minimal interference with biochemical processes, making it ideal for applications ranging from cell culture to molecular biology techniques. The buffering action of PIPES sodium salt centers around its ability to maintain a consistent pH in the range of 6.1 to 7.5, crucial for maintaining the integrity and functionality of biological molecules. Its mechanism involves the absorption and release of hydrogen ions (protons), which prevents significant pH changes in the solution, even when acids or bases are added or when biochemical reactions generate acidic or basic products. This characteristic is particularly important in environments such as cell culture, where pH stability is essential for cell health and function. Additionally, PIPES sodium salt is often used in the preparation of buffers for electrophoresis and other enzyme assays, where it supports optimal enzyme activity and stability by maintaining a stable pH. Its role in research has been instrumental in ensuring that experimental conditions remain constant, thereby enhancing the reliability and reproducibility of scientific outcomes.
PIPES sodium salt (CAS 10010-67-0) References
- The influence of the buffer on maintenance of tissue lipid in specimens for scanning electron microscopy. | Schiff, RI. and Gennaro, JF. 1979. Scan Electron Microsc. 449-58. PMID: 118516
- Hydrogen ion buffers for biological research. | Good, NE., et al. 1966. Biochemistry. 5: 467-77. PMID: 5942950
- Effects of NaOH-PIPES buffer used in aldehyde fixative on alkaline phosphatase activity in rat hepatocytes. | Yamamoto, K. and Ogawa, K. 1983. Histochemistry. 77: 339-51. PMID: 6305886
- Buffers for enzymes. | Blanchard, JS. 1984. Methods Enzymol. 104: 404-14. PMID: 6717292
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
PIPES sodium salt, 100 g | sc-296097 | 100 g | $163.00 | |||
PIPES sodium salt, 500 g | sc-296097A | 500 g | $595.00 | |||
PIPES sodium salt, 1 kg | sc-296097B | 1 kg | $1122.00 |
