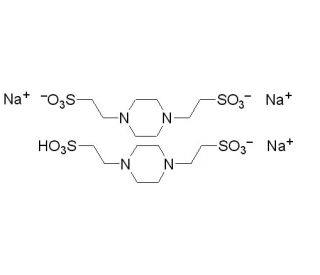
PIPES, Sesquisodium Salt 的分子结构, CAS编号: 100037-69-2
PIPES, Sesquisodium Salt (CAS 100037-69-2)
备用名:
1,4-Piperazinediethanesulfonic acid sesquisodium salt; Piperazine-1,4-bis(2-ethanesulfonic acid) sesquisodium salt
应用:
PIPES, Sesquisodium Salt 是 PIPES 钠盐,是一种常用的缓冲剂
CAS号码:
100037-69-2
分子量:
335.34
分子式:
C8H16.5N2O6S2•1.5Na
仅供科研使用。不可用于诊断或治疗。
* 参考分析证明 大量特定数据 (包括水 含量).
快捷链接
订购信息
描述
技术信息
安全信息
安全技术说明书和分析证明书
PIPES 或哌嗪-N,N'-双(2-乙磺酸)是一种齐聚物缓冲化合物,广泛用于生化和生物物理研究。它在 6.1 到 7.5 的 pH 值范围内具有出色的缓冲能力,因此特别适合在对 pH 值变化敏感的环境中保持 pH 值的稳定性。PIPES 的结构包括一个哌嗪环和两个乙磺酸基团,使其具有接受或捐赠质子的能力,从而即使在不同的实验条件下也能稳定溶液的 pH 值。在涉及酶反应、细胞培养系统和其他生物检测的研究中,pH 值的微小偏差都会严重影响分子的生物活性和稳定性,因此这一特性至关重要。在细胞生物学中,PIPES 主要用于缓冲细胞培养基,因为它与细胞成分的相互作用极小,并能维持与生理相关的环境。此外,PIPES 还被用于各种分子生物学方案,包括核酸提取和电泳,其卓越的缓冲作用可确保核酸在操作和分析过程中的完整性和稳定性。PIPES 的离子强度低,与金属离子的结合力极小,因此也是涉及金属敏感过程的研究的理想选择,从而为需要精确控制生化条件的各种科学研究提供了支持。
PIPES, Sesquisodium Salt (CAS 100037-69-2) 参考文献
- 缓冲液对保持扫描电子显微镜样本中组织脂质的影响。 | Schiff, RI. and Gennaro, JF. 1979. Scan Electron Microsc. 449-58. PMID: 118516
- 一纳米精度的荧光成像:分子马达的体外和体内研究。 | Hoffman, MT., et al. 2011. Methods Mol Biol. 778: 33-56. PMID: 21809199
- 在生物缓冲液中合成尺寸和形态随缓冲液变化的金纳米粒子。 | Ahmed, SR., et al. 2016. Nanoscale Res Lett. 11: 65. PMID: 26847691
- 产甲烷古细菌中 RuBisCO 介导的碳代谢途径。 | Kono, T., et al. 2017. Nat Commun. 8: 14007. PMID: 28082747
- 四个依赖叶酸的蛋氨酸合成酶家族。 | Price, MN., et al. 2021. PLoS Genet. 17: e1009342. PMID: 33534785
- 用于癌细胞端粒酶活性成像的 DNA 四面体对接组件。 | Zhang, R., et al. 2022. Anal Chim Acta. 1193: 339395. PMID: 35058008
- 利用吉布森组装技术高效生成用于蛋白质配体筛选的全基因组文库。 | Sternlieb, T., et al. 2022. Bio Protoc. 12: PMID: 36532687
- 用于生物研究的氢离子缓冲器。 | Good, NE., et al. 1966. Biochemistry. 5: 467-77. PMID: 5942950
- 醛固定液中使用的 NaOH-PIPES 缓冲液对大鼠肝细胞碱性磷酸酶活性的影响。 | Yamamoto, K. and Ogawa, K. 1983. Histochemistry. 77: 339-51. PMID: 6305886
- 酶的缓冲液 | Blanchard, JS. 1984. Methods Enzymol. 104: 404-14. PMID: 6717292
订购信息
| 产品名称 | 产品编号 | 规格 | 价格 | 数量 | 收藏夹 | |
PIPES, Sesquisodium Salt, 100 g | sc-216100 | 100 g | $102.00 | |||
PIPES, Sesquisodium Salt, 1 kg | sc-216100A | 1 kg | $714.00 |
