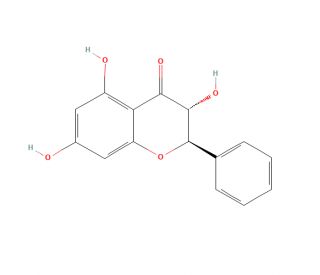
Molecular structure of Pinobanksin, CAS Number: 548-82-3
Pinobanksin (CAS 548-82-3)
Alternate Names:
(2R,3R)-3,5,7-Trihydroxy-2-phenyl-2,3-dihydro-4H-chromen-4-one; (2R,3R)-3,5,7-Trihydroxy-2-phenyl-chroman-4-one
CAS Number:
548-82-3
Molecular Weight:
272.25
Molecular Formula:
C15H12O5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Pinobanksin is extensively used in research focused on flavonoids and their role in plant defense mechanisms. This compound is studied for its antioxidant properties, helping scientists understand how plants protect themselves from oxidative stress. In biochemical research, Pinobanksin is applied in studies to elucidate its interaction with various enzymes involved in the biosynthesis of flavonoids. Additionally, it serves as a model compound in the study of the metabolic pathways of secondary metabolites in plants. Researchers also utilize Pinobanksin to investigate its potential impact on the stability and expression of certain genes involved in plant immunity and resilience.
Pinobanksin (CAS 548-82-3) References
- Identification of metabolites in plasma and urine of Uruguayan propolis-treated rats. | Kumazawa, S., et al. 2004. J Agric Food Chem. 52: 3083-8. PMID: 15137857
- Evaluation of propolis polyphenols absorption in humans by liquid chromatography/tandem mass spectrometry. | Gardana, C., et al. 2007. Rapid Commun Mass Spectrom. 21: 3849-54. PMID: 17979105
- Effect of Ethanol/Water Solvents on Phenolic Profiles and Antioxidant Properties of Beijing Propolis Extracts. | Sun, C., et al. 2015. Evid Based Complement Alternat Med. 2015: 595393. PMID: 26351514
- Apoptotic induction by pinobanksin and some of its ester derivatives from Sonoran propolis in a B-cell lymphoma cell line. | Alday, E., et al. 2015. Chem Biol Interact. 242: 35-44. PMID: 26367700
- Honey flavonoids inhibit hOATP2B1 and hOATP1A2 transporters and hOATP-mediated rosuvastatin cell uptake in vitro. | Navrátilová, L., et al. 2018. Xenobiotica. 48: 745-755. PMID: 28745105
- Theoretical studies on the antioxidant activity of pinobanksin and its ester derivatives: Effects of the chain length and solvent. | Zheng, YZ., et al. 2018. Food Chem. 240: 323-329. PMID: 28946279
- Effect of propolis phenolic compounds on free fatty acid receptor 4 activation. | Cho, H., et al. 2020. Food Sci Biotechnol. 29: 579-584. PMID: 32296569
- Natural products isolation studies of the paleoendemic plant species Nothofagus gunnii and Nothofagus cunninghamii. | Gyeltshen, T., et al. 2022. Fitoterapia. 156: 105088. PMID: 34798163
- The Profile of Polyphenolic Compounds, Contents of Total Phenolics and Flavonoids, and Antioxidant and Antimicrobial Properties of Bee Products. | Sawicki, T., et al. 2022. Molecules. 27: PMID: 35209088
- A comprehensive chemical analysis of New Zealand honeydew honey. | Jonathan Chessum, K., et al. 2022. Food Res Int. 157: 111436. PMID: 35761676
- The Antioxidant and Anti-Inflammatory Effects of Flavonoids from Propolis via Nrf2 and NF-κB Pathways. | Xu, W., et al. 2022. Foods. 11: PMID: 36010439
- Biological Activity and Chemical Composition of Propolis from Various Regions of Poland. | Woźniak, M., et al. 2022. Molecules. 28: PMID: 36615334
- Correlation between Chemical Profile of Georgian Propolis Extracts and Their Activity against Helicobacter pylori. | Widelski, J., et al. 2023. Molecules. 28: PMID: 36771040
- Antimicrobial, Antibiofilm and Toxicological Assessment of Propolis. | Queiroga, MC., et al. 2023. Antibiotics (Basel). 12: PMID: 36830258
- Pinobanksin inhibits peroxidation of low density lipoprotein and it has electron donor properties reducing alpha-tocopherol radicals. | Ondrias, K., et al. 1997. Pharmazie. 52: 566-7. PMID: 9266597
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pinobanksin, 2 mg | sc-476233 | 2 mg | $300.00 |
