Picrotin (CAS 21416-53-5)
QUICK LINKS
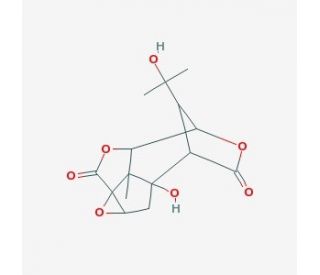
Picrotin, a natural picrotoxane, exhibits antagonistic activity against glycine receptors (GlyRs). It specifically targets α1 and α2 homodimeric GlyRs, with IC50 values of 57 µM and 117 µM, respectively. Additionally, it inhibits α3 homodimeric GlyRs. Notably, Picrotin does not display inhibitory effects on γ-aminobutyric acid (GABA) type A and type C receptors. This compound naturally occurs in picrotoxin, a plant-derived poison, where it exists in equimolar quantities with picrotoxinin. As an analytical standard, Picrotin is provided with high chromatographic purity, serving as a reference material for qualitative determination. Structurally, it is an organic heteropentacyclic compound formed from picrotoxinin through the addition of water to the olefinic double bond, resulting in a tertiary alcohol. Picrotin represents the less toxic component of picrotoxin, devoid of GABA activity. Functionally, it acts as a plant metabolite and belongs to the group of organic heteropentacyclic compounds, possessing characteristics such as being an epoxide, tertiary alcohol, gamma-lactone, diol, and picrotoxane sesquiterpenoid. Its origin can be traced back to picrotoxinin. Picrotin′s selective antagonism of glycine receptors, coupled with its distinct chemical properties, highlights its importance as a compound of interest in various research fields, particularly in understanding receptor interactions and conducting qualitative determinations.
Picrotin (CAS 21416-53-5) References
- Subunit-specific action of an anticonvulsant thiobutyrolactone on recombinant glycine receptors involves a residue in the M2 membrane-spanning region. | Steinbach, JH., et al. 2000. Mol Pharmacol. 58: 11-7. PMID: 10860922
- NOESY on neurotoxins: NMR and conformational assignments of picrotoxins. | Perry, NB., et al. 2001. Phytochem Anal. 12: 69-72. PMID: 11704965
- Benzodiazepines block alpha2-containing inhibitory glycine receptors in embryonic mouse hippocampal neurons. | Thio, LL., et al. 2003. J Neurophysiol. 90: 89-99. PMID: 12660352
- Mechanisms for picrotoxinin and picrotin blocks of alpha2 homomeric glycine receptors. | Wang, DS., et al. 2007. J Biol Chem. 282: 16016-35. PMID: 17405877
- Glycine receptor subunit composition alters the action of GABA antagonists. | Li, P. and Slaughter, M. 2007. Vis Neurosci. 24: 513-21. PMID: 17659095
- A proposed structural basis for picrotoxinin and picrotin binding in the glycine receptor pore. | Yang, Z., et al. 2007. J Neurochem. 103: 580-9. PMID: 17714449
- Ginkgolide B and bilobalide block the pore of the 5-HT₃receptor at a location that overlaps the picrotoxin binding site. | Thompson, AJ., et al. 2011. Neuropharmacology. 60: 488-95. PMID: 21059362
- Gating effects on picrotin block of glycine receptors. | Li, P. and Slaughter, MM. 2012. Neuroreport. 23: 1017-20. PMID: 23079787
- Plant regeneration, genetic fidelity, and active ingredient content of encapsulated hairy roots of Picrorhiza kurrooa Royle ex Benth. | Rawat, JM., et al. 2013. Biotechnol Lett. 35: 961-8. PMID: 23397268
- GABA receptors on the cell-body membrane of an identified insect motor neuron. | Sattelle, DB., et al. 1988. Proc R Soc Lond B Biol Sci. 232: 443-56. PMID: 2451252
- Simultaneous determination of the two components of picrotoxin in serum by reversed-phase high-performance liquid chromatography with application to a pharmacokinetic study in rats. | Soto-Otero, R., et al. 1989. J Pharm Biomed Anal. 7: 369-75. PMID: 2488637
- Untargeted serum metabolomic profiling for early detection of Schistosoma mekongi infection in mouse model. | Chienwichai, P., et al. 2022. Front Cell Infect Microbiol. 12: 910177. PMID: 36061860
- Mutations affecting the glycine receptor agonist transduction mechanism convert the competitive antagonist, picrotoxin, into an allosteric potentiator. | Lynch, JW., et al. 1995. J Biol Chem. 270: 13799-806. PMID: 7775436
- Picrotoxin as a potent inducer of rat hepatic cytochrome P450, CYP2B1 and CYP2B2. | Yamada, H., et al. 1993. Biochem Pharmacol. 45: 1783-9. PMID: 8494537
- The human glycine receptor beta subunit: primary structure, functional characterisation and chromosomal localisation of the human and murine genes. | Handford, CA., et al. 1996. Brain Res Mol Brain Res. 35: 211-9. PMID: 8717357
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Picrotin, 250 mg | sc-228947 | 250 mg | $150.00 |
