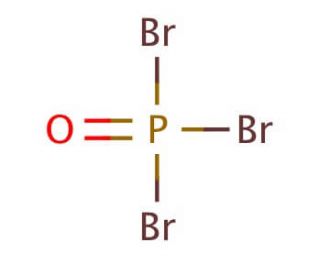
Molecular structure of Phosphorus(V) oxybromide, CAS Number: 7789-59-5
Phosphorus(V) oxybromide (CAS 7789-59-5)
Alternate Names:
Phosphorus oxide bromide; Phosphoryl bromide
CAS Number:
7789-59-5
Molecular Weight:
286.69
Molecular Formula:
Br3OP
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
Available in US only.
Available in US only.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Phosphorus(V) oxybromide functions as a powerful brominating agent in various chemical reactions. Its mechanism of action involves the transfer of bromine atoms to organic compounds, leading to the formation of new carbon-bromine bonds. This process is particularly useful in the synthesis of agrochemicals, as well as in the modification of organic molecules. Phosphorus(V) oxybromide′s reactivity allows for the selective introduction of bromine atoms into specific positions within a molecule, enabling the creation of diverse chemical structures.
Phosphorus(V) oxybromide (CAS 7789-59-5) References
- Expeditious Lead Optimization of Isoxazole-Containing Influenza A Virus M2-S31N Inhibitors Using the Suzuki-Miyaura Cross-Coupling Reaction. | Li, F., et al. 2017. J Med Chem. 60: 1580-1590. PMID: 28182419
- Discovery of Potent 2-Aryl-6,7-dihydro-5 H-pyrrolo[1,2- a]imidazoles as WDR5-WIN-Site Inhibitors Using Fragment-Based Methods and Structure-Based Design. | Wang, F., et al. 2018. J Med Chem. 61: 5623-5642. PMID: 29889518
- Discovery of QCA570 as an Exceptionally Potent and Efficacious Proteolysis Targeting Chimera (PROTAC) Degrader of the Bromodomain and Extra-Terminal (BET) Proteins Capable of Inducing Complete and Durable Tumor Regression. | Qin, C., et al. 2018. J Med Chem. 61: 6685-6704. PMID: 30019901
- Thermo-Responsive Fluorescent Polymers with Diverse LCSTs for Ratiometric Temperature Sensing through FRET. | Ding, Z., et al. 2018. Polymers (Basel). 10: PMID: 30966318
- Synthesis of distal and proximal fleximer base analogues and evaluation in the nucleocapsid protein of HIV-1. | Ku, T., et al. 2019. Bioorg Med Chem. 27: 2883-2892. PMID: 31126822
- Diversification of Nucleophile-Intercepted Beckmann Fragmentation Products and Related Density Functional Theory Studies. | Lowder, LL., et al. 2020. J Org Chem. 85: 11396-11408. PMID: 32786611
- Identification of 2,4-Disubstituted Imidazopyridines as Hemozoin Formation Inhibitors with Fast-Killing Kinetics and In Vivo Efficacy in the Plasmodium falciparum NSG Mouse Model. | Horatscheck, A., et al. 2020. J Med Chem. 63: 13013-13030. PMID: 33103428
- Multisubstituted pyrimidines effectively inhibit bacterial growth and biofilm formation of Staphylococcus aureus. | Provenzani, R., et al. 2021. Sci Rep. 11: 7931. PMID: 33846401
- BAY-069, a Novel (Trifluoromethyl)pyrimidinedione-Based BCAT1/2 Inhibitor and Chemical Probe. | Günther, J., et al. 2022. J Med Chem. 65: 14366-14390. PMID: 36261130
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Phosphorus(V) oxybromide, 25 g | sc-250714 | 25 g | $224.00 | |||
| US: Only available in the US | ||||||
