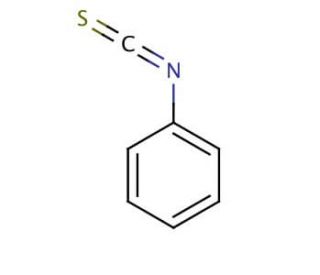
Molecular structure of Phenyl isothiocyanate, CAS Number: 103-72-0
Phenyl isothiocyanate (CAS 103-72-0)
Alternate Names:
PITC
Application:
Phenyl isothiocyanate is which inhibits NNK-induced tumorigenesis
CAS Number:
103-72-0
Purity:
≥97%
Molecular Weight:
135.19
Molecular Formula:
C7H5NS
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Phenyl isothiocyanate is an inhibitor of nicotine-derived nitrosamine ketone (NNK)-induced lung tumorigenesis. Phenyl isothiocyanate, derived from benzenethiol, finds versatile applications in various compound syntheses. It serves as a component in the production of dyes, fragrances, and other compounds. Moreover, phenyl isothiocyanate plays a role in synthesizing diverse polymers like polyurethanes, polyesters, and polyimides. Additionally, it has been utilized in the creation of catalysts, such as palladium and nickel catalysts.
Phenyl isothiocyanate (CAS 103-72-0) References
- 1,3-Heterocumulene-to-alkyne. | Fabian, J., et al. 2000. J Org Chem. 65: 8940-7. PMID: 11149836
- Photodissociation tandem mass spectrometry at 266 nm of an aliphatic peptide derivatized with phenyl isothiocyanate and 4-sulfophenyl isothiocyanate. | Oh, JY., et al. 2005. Rapid Commun Mass Spectrom. 19: 1283-8. PMID: 15838844
- Allyl isothiocyanate (AITC) and phenyl isothiocyanate (PITC) inhibit tumour-specific angiogenesis by downregulating nitric oxide (NO) and tumour necrosis factor-alpha (TNF-alpha) production. | Thejass, P. and Kuttan, G. 2007. Nitric Oxide. 16: 247-57. PMID: 17097904
- Inhibition of endothelial cell differentiation and proinflammatory cytokine production during angiogenesis by allyl isothiocyanate and phenyl isothiocyanate. | Thejass, P. and Kuttan, G. 2007. Integr Cancer Ther. 6: 389-99. PMID: 18048887
- Antibacterial activity of phenyl isothiocyanate on Escherichia coli and Staphylococcus aureus. | Abreu, AC., et al. 2013. Med Chem. 9: 756-61. PMID: 22974327
- A convenient synthesis of 2-imino-1,3-thiazine-4-one derivatives from the reaction of primary amines and phenyl isothiocyanate in the presence of acryloyl chloride. | Zolali, A., et al. 2014. Comb Chem High Throughput Screen. 17: 610-3. PMID: 24628280
- Identification of a novel food-derived collagen peptide, hydroxyprolyl-glycine, in human peripheral blood by pre-column derivatisation with phenyl isothiocyanate. | Shigemura, Y., et al. 2011. Food Chem. 129: 1019-24. PMID: 25212331
- Insertion of phenyl isothiocyanate into a P-P bond of a nickel-substituted bicyclo[1.1.0]tetraphosphabutane. | Pelties, S., et al. 2016. Chem Commun (Camb). 52: 6601-4. PMID: 27111495
- Superoxide anion measurement by sulfonated phenyl isothiocyanate cytochrome c. | Morel, F., et al. 1988. Biochem Biophys Res Commun. 156: 1175-81. PMID: 2847727
- Rotational Spectra and Structures of Phenyl Isocyanate and Phenyl Isothiocyanate. | Sun, W., et al. 2019. J Phys Chem A. 123: 2351-2360. PMID: 30817149
- Rapid analysis of phenyl isothiocyanate derivatives of amino acids present in Czech meads. | Klikarová, J., et al. 2021. J Chromatogr A. 1644: 462134. PMID: 33848765
- Persistence Enhancement of a Promising Tick Repellent, Benzyl Isothiocyanate, by Yeast Microcarriers. | Kim, HJ., et al. 2021. Molecules. 26: PMID: 34833912
- Bioavailability of Tauropine After Oral Ingestion in Mouse. | Ito, T., et al. 2022. Adv Exp Med Biol. 1370: 137-142. PMID: 35882789
- Four-week toxicity study of phenyl isothiocyanate in rats. | Speijers, GJ., et al. 1985. Food Chem Toxicol. 23: 1015-7. PMID: 4076929
- Chemopreventive activity of thiol conjugates of isothiocyanates for lung tumorigenesis. | Jiao, D., et al. 1997. Carcinogenesis. 18: 2143-7. PMID: 9395214
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Phenyl isothiocyanate, 50 g | sc-204842 | 50 g | $68.00 | |||
Phenyl isothiocyanate, 100 g | sc-204842A | 100 g | $104.00 |
