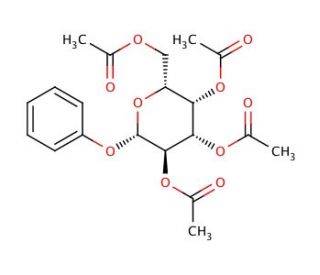
Phenyl-2,3,4,6-tetra-O-acetyl-β-D-galactopyranoside (CAS 2872-72-2)
QUICK LINKS
Phenyl-2,3,4,6-tetra-O-acetyl-β-D-galactopyranoside, a synthetic derivative of β-D-galactopyranoside, has garnered significant attention in research due to its utility as a substrate for enzymatic assays and glycosylation reactions. In enzymatic studies, this compound serves as a substrate for various glycosidases, enzymes that catalyze the hydrolysis of glycosidic bonds. Researchers exploit its structural properties to investigate the substrate specificity and kinetic parameters of glycosidases, crucial for understanding their biological functions and developing enzyme inhibitors. Additionally, Phenyl-2,3,4,6-tetra-O-acetyl-β-D-galactopyranoside is employed as a glycosyl donor in glycosylation reactions, where it acts as a sugar component in the synthesis of complex carbohydrates or glycoconjugates. This chemical is particularly valuable in the synthesis of oligosaccharides and glycopeptides for glycobiology research, enabling the construction of structurally diverse glycan libraries for studying carbohydrate-protein interactions, cell signaling, and immunology. Moreover, its acetyl groups confer stability and solubility, facilitating its use in organic synthesis and chemical biology studies. Overall, Phenyl-2,3,4,6-tetra-O-acetyl-β-D-galactopyranoside plays a pivotal role in elucidating glycoside hydrolase mechanisms and advancing research in glycoscience and chemical glycobiology.
Phenyl-2,3,4,6-tetra-O-acetyl-β-D-galactopyranoside (CAS 2872-72-2) References
- Phenyl galactopyranosides–13C CPMAS NMR and conformational analysis using genetic algorithm[J]. | Wałejko P, Paradowska K, Bukowicki J. 2015. Chemical Physics,., 457:: 43-50.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Phenyl-2,3,4,6-tetra-O-acetyl-β-D-galactopyranoside, 1 g | sc-391651 | 1 g | $300.00 |
