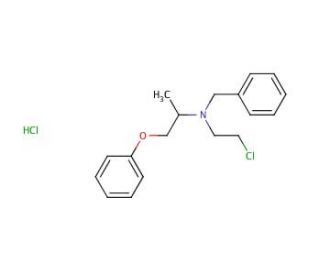
Phenoxybenzamine (CAS 63-92-3)
See product citations (4)
QUICK LINKS
Phenoxybenzamine is used in physiological research, particularly for its capacity to irreversibly block alpha-adrenergic receptors. This blockade is for studies focused on the sympathetic nervous system and its role in regulating vascular tone and blood pressure. Phenoxybenzamine′s ability to induce vasodilation makes it useful for investigating the pathophysiology of diseases characterized by excessive vasoconstriction and hypertension. Additionally, it is used in research to explore the mechanisms of catecholamine release and receptor dynamics under various physiological and pathological conditions. Furthermore, Phenoxybenzamine is utilized in studies that examine the effects of adrenergic receptor inhibition on smooth muscle function, contributing to a broader understanding of autonomic regulation.
Phenoxybenzamine (CAS 63-92-3) References
- Phenoxybenzamine in the management of neurogenic vesical dysfunction. | Mobley, DF. 1976. J Urol. 116: 737-8. PMID: 1003641
- Phenoxybenzamine is more effective and less harmful than papaverine in the prevention of radial artery vasospasm. | Dipp, MA., et al. 2001. Eur J Cardiothorac Surg. 19: 482-6. PMID: 11306317
- Phenoxybenzamine binding reveals the helical orientation of the third transmembrane domain of adrenergic receptors. | Frang, H., et al. 2001. J Biol Chem. 276: 31279-84. PMID: 11395517
- Urethral pressure profile and hemodynamic effects of phenoxybenzamine and prazosin in non-sedated male beagle dogs. | Fischer, JR., et al. 2003. Can J Vet Res. 67: 30-8. PMID: 12528826
- Recent developments in the therapy of phaeochromocytoma. | Chew, SL. 2004. Expert Opin Investig Drugs. 13: 1579-83. PMID: 15566315
- Treatment of complex regional pain syndrome type I with oral phenoxybenzamine: rationale and case reports. | Inchiosa, MA. and Kizelshteyn, G. 2008. Pain Pract. 8: 125-32. PMID: 18194348
- Phenoxybenzamine treatment can lead to loss of endothelial cell viability. | Pai, KR., et al. 2008. Interact Cardiovasc Thorac Surg. 7: 916-8. PMID: 18573847
- Inhibition of nitric oxide-activated guanylyl cyclase by calmodulin antagonists. | James, LR., et al. 2009. Br J Pharmacol. 158: 1454-64. PMID: 19845679
- Phenoxybenzamine is neuroprotective in a rat model of severe traumatic brain injury. | Rau, TF., et al. 2014. Int J Mol Sci. 15: 1402-17. PMID: 24447929
- Comparison of phenoxybenzamine and doxazosin in perioperative management of patients with pheochromocytoma. | Malec, K., et al. 2017. Kardiol Pol. 75: 1192-1198. PMID: 28715066
- Anti-tumor activity of phenoxybenzamine and its inhibition of histone deacetylases. | Inchiosa, MA. 2018. PLoS One. 13: e0198514. PMID: 29897996
- Comparison of Preoperative Alpha-blockade for Resection of Paraganglioma and Pheochromocytoma. | Zhu, CY., et al. 2022. Endocr Pract. 28: 889-896. PMID: 35809774
- Interaction of alpha adrenergic antagonists with calmodulin. | Earl, CQ., et al. 1984. Life Sci. 35: 525-34. PMID: 6146911
- Phenoxybenzamine for the treatment of severe nonresponsive diarrhea in the horse. | Hood, DM., et al. 1982. J Am Vet Med Assoc. 180: 758-62. PMID: 7085456
- In vivo evidence for the generation of a glucocorticoid receptor-heat shock protein-90 complex incapable of binding hormone by the calmodulin antagonist phenoxybenzamine. | Ning, YM. and Sánchez, ER. 1996. Mol Endocrinol. 10: 14-23. PMID: 8838141
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Phenoxybenzamine, 200 mg | sc-3581 | 200 mg | $23.00 | |||
Phenoxybenzamine, 1 g | sc-3581A | 1 g | $77.00 |
