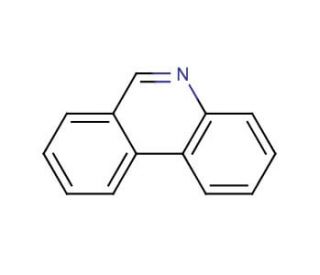
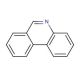
Phenanthridine (CAS 229-87-8)
QUICK LINKS
Phenanthridine is a heterocyclic organic compound that serves as a key scaffold in the synthesis of various complex molecules used in materials science and organic electronics. Its planar structure and electronic properties make it an interesting candidate for the design of organic semiconductors and light-emitting diodes (LEDs), where it is investigated for its charge-transport and photophysical behaviors. In the field of supramolecular chemistry, phenanthridine is studied for its ability to intercalate with DNA, which is useful in understanding the interactions between small molecules and biopolymers. Additionally, it acts as a ligand in coordination chemistry, forming complexes with metals, which are then explored for catalysis and as potential sensors.
Phenanthridine (CAS 229-87-8) References
- Synthesis of phenanthridine spiropyrans and studies of their effects on G-quadruplex DNA. | Livendahl, M., et al. 2017. Org Biomol Chem. 15: 3265-3275. PMID: 28349141
- Metabolites of phenanthridine formed by rat liver homogenate. | LaVoie, EJ., et al. 1985. Drug Metab Dispos. 13: 71-5. PMID: 2858381
- Efficient synthesis of new phenanthridine Wnt/β-catenin signaling pathway agonists. | Chen, D., et al. 2018. Eur J Med Chem. 157: 1491-1499. PMID: 30282321
- Structurally Simple Phenanthridine Analogues Based on Nitidine and Their Antitumor Activities. | Qin, SQ., et al. 2019. Molecules. 24: PMID: 30691109
- Phenanthridine derivatives as promising new anticancer agents: synthesis, biological evaluation and binding studies. | Azad, I., et al. 2020. Future Med Chem. 12: 709-739. PMID: 32208986
- Phenanthridine Sulfonamide Derivatives as Potential DPP-IV Inhibitors: Design, Synthesis and Biological Evaluation. | Khalaf, RA., et al. 2022. Curr Comput Aided Drug Des. 18: 9-25. PMID: 33030135
- Phenanthridine Derivative Host Heat Shock Cognate 70 Down-Regulators as Porcine Epidemic Diarrhea Virus Inhibitors. | Chen, DZ., et al. 2021. J Nat Prod. 84: 1175-1184. PMID: 33760626
- Is the cytotoxic activity of phenanthriplatin dependent on the specific size of the phenanthridine ligand π system? | Scoditti, S., et al. 2021. J Inorg Biochem. 219: 111447. PMID: 33798829
- In-Cell Generation of Anticancer Phenanthridine Through Bioorthogonal Cyclization in Antitumor Prodrug Development. | Maslah, H., et al. 2021. Angew Chem Int Ed Engl. 60: 24043-24047. PMID: 34487611
- Novel phenanthridine amide analogs as potential anti-leishmanial agents: In vitro and in silico insights. | Nandikolla, A., et al. 2021. Bioorg Chem. 117: 105414. PMID: 34655843
- Novel nucleocapsid protein-targeting phenanthridine inhibitors of SARS-CoV-2. | Wang, YT., et al. 2022. Eur J Med Chem. 227: 113966. PMID: 34749200
- Electrochemical Decarboxylative Cyclization of α-Amino-Oxy Acids to Access Phenanthridine Derivatives. | Zhan, Y., et al. 2022. Chem Asian J. 17: e202101388. PMID: 35043595
- Design, synthesis and evaluation of novel phenanthridine triazole analogs as potential antileishmanial agents. | Nandikolla, A., et al. 2022. Future Med Chem. 14: 867-880. PMID: 35642458
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Phenanthridine, 1 g | sc-215698 | 1 g | $50.00 |
