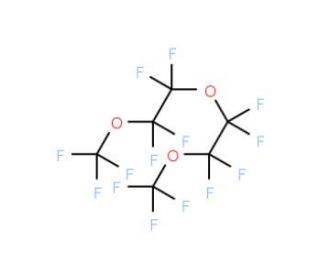
Perfluoro(diethylene glycol dimethyl ether) (CAS 40891-99-4)
QUICK LINKS
Perfluoro(diethylene glycol dimethyl ether) is a compound that has piqued the interest of researchers in the fields of materials science and chemical engineering due to its unique perfluorinated ether structure. This compound is widely used in studies exploring its properties as a high-performance solvent, particularly in applications that require chemical inertness, thermal stability, and low surface tension. In the area of electrochemistry, Perfluoro(diethylene glycol dimethyl ether) is investigated for its potential use in electrolytes for lithium batteries, where it can contribute to the stability and ionic conductivity of the battery′s electrolyte. Its utility in the synthesis of fluorinated compounds is also of interest due to its ability to dissolve reactants that are typically challenging to handle in non-fluorinated environments. Furthermore, Perfluoro(diethylene glycol dimethyl ether) is used in the development of advanced lubricants and coatings, where its fluorinated nature imparts excellent lubricity and resistance to harsh conditions. Researchers also examine its role in gas separation technologies, leveraging its selective solvation properties to facilitate the separation of gases like oxygen and nitrogen.
Perfluoro(diethylene glycol dimethyl ether) (CAS 40891-99-4) References
- Strong fragmentation processes driven by low energy electron attachment to various small perfluoroether molecules. | Mitterdorfer, C., et al. 2011. Int J Mass Spectrom. 306: 63-69. PMID: 21977005
- Effects of perfluorocarbon gases on the size and stability characteristics of phospholipid-coated microbubbles: osmotic effect versus interfacial film stabilization. | Szíjjártó, C., et al. 2012. Langmuir. 28: 1182-9. PMID: 22176688
- Electron ionization of different large perfluoroethers embedded in ultracold helium droplets: effective freezing of short-lived decomposition intermediates. | Bartl, P., et al. 2013. Rapid Commun Mass Spectrom. 27: 298-304. PMID: 23239377
- Glymes as Versatile Solvents for Chemical Reactions and Processes: from the Laboratory to Industry. | Tang, S. and Zhao, H. 2014. RSC Adv. 4: 11251-11287. PMID: 24729866
- Perfluoropolyethers: Development of an All-Atom Force Field for Molecular Simulations and Validation with New Experimental Vapor Pressures and Liquid Densities. | Black, JE., et al. 2017. J Phys Chem B. 121: 6588-6600. PMID: 28557461
- Dissolution of multicomponent microbubbles in the bloodstream: 2. Experiment. | Kabalnov, A., et al. 1998. Ultrasound Med Biol. 24: 751-60. PMID: 9695278
- On the kinetics of thermal electron attachment to perfluoroethers | J. Kopyra a, J. Wnorowska a, W. Barszczewska a, S. Karolczak b, I. Szamrej a. 2012. Chemical Physics Letters. 519–520: 25-28.
- Perfluoroalkyl chains as tools for film surface nano-patterning and soft microbubble engineering and decoration | Marie Pierre Krafft. 2014. Journal of the Taiwan Institute of Chemical Engineers. 45: 3069-3074.
- Kinetics of low energy electron attachment to some fluorinated alcohols in the gas phase | K. Wnorowski, J. Wnorowska, J. Kopyra, B. Michalczuk, I. Szamrej, W. Barszczewska. 2014. Chemical Physics Letters. 591: 282-286.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Perfluoro(diethylene glycol dimethyl ether), 5 g | sc-264028 | 5 g | $166.00 | |||
Perfluoro(diethylene glycol dimethyl ether), 10 g | sc-264028A | 10 g | $292.00 |
