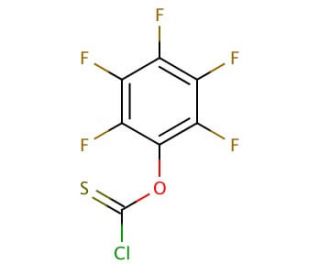
Pentafluorophenyl chlorothionoformate (CAS 135192-53-9)
QUICK LINKS
Pentafluorophenyl chlorothionoformate, known as PFP-CTF, is a remarkably versatile and reactive reagent, finding extensive use in diverse scientific research applications. Its capabilities are vast, enabling the introduction of fluorine atoms into organic molecules, the synthesis of fluorinated compounds, heterocyclic compounds, and peptides. Additionally, it plays a role in pharmaceutical synthesis and contributes to the creation of biocompatible materials, like polymers and hydrogels. Moreover, Pentafluorophenyl chlorothionoformate has been instrumental in exploring the biochemical effects of various compounds. The reaction involving Pentafluorophenyl chlorothionoformate is initiated via an SN2 mechanism, where the chlorine atom displaces the hydroxyl group of pentafluorophenol. This results in a highly reactive and versatile product, well-suited for a wide range of organic synthesis reactions. With its exceptional properties, Pentafluorophenyl chlorothionoformate remains an indispensable tool in the pursuit of advancements in scientific research and various industries.
Pentafluorophenyl chlorothionoformate (CAS 135192-53-9) References
- Regioselective synthesis of beta-N1- and beta-N3-alloxazine nucleosides. | Wang, Z. and Rizzo, CJ. 2000. Org Lett. 2: 227-30. PMID: 10814288
- Tuning the acceptors in catalyzed cyclizations initiated by allenes. Silylstannylation/cyclization of allene-aldehydes for synthesis of polyalkylated indolizidines including 223A congeners. | Kumareswaran, R., et al. 2004. J Org Chem. 69: 9151-8. PMID: 15609949
- Synthesis and enzymatic evaluation of modified acceptors of recombinant blood group A and B glycosyltransferases. | Mukherjee, A., et al. 2000. Carbohydr Res. 326: 1-21. PMID: 16001502
- Synthesis of 3'-C-substituted thymidine derivatives by free-radical techniques: scope and limitations. | Horton, D., et al. 2007. Carbohydr Res. 342: 259-67. PMID: 17173879
- Diastereoselective Total Synthesis of (-)-Galiellalactone. | Kim, T., et al. 2015. J Org Chem. 80: 12193-200. PMID: 26544529
- Total Syntheses of (+)-Peniciketals A-B and (-)-Diocollettines A Exploiting a Photoisomerization/Cyclization Union Protocol. | Deng, Y., et al. 2021. J Org Chem. 86: 13583-13597. PMID: 34510901
- Recognition of Dimethylarginine Analogues by Tandem Tudor Domain Protein Spindlin1. | Porzberg, MRB., et al. 2022. Molecules. 27: PMID: 35164245
- A stereospecific route to 2-deoxy-.beta.-glycosides | Jacquelyn Gervay and S. Danishefsky. 1991. J. Org. Chem., 56, 18,: 5448–5451.
- Total Synthesis of (+)-Aloperine. Use of a Nitrogen-Bound Silicon Tether in an Intramolecular Diels−Alder Reaction | Arthur D. Brosius, Larry E. Overman, and Lothar Schwink. 1999,. J. Am. Chem. Soc. 121, 4,: 700–709.
- Convenient Synthesis of 2-Deoxy-D-Glucofuranosides | José Molina Arévalo and & Claire Simons. 1999 -. Journal of Carbohydrate Chemistry. Volume 18, Issue 5: Pages 535-544.
- Mapping the Active Site of the Bacterial Enzyme LpxC Using Novel Carbohydrate‐Based Hydroxamic Acid InhibitorsFootnote* | Xuechen Li, et al. 2005 -. Journal of Carbohydrate Chemistry. Volume 24, Issue 4-6: Pages 583-609.
- The invention of radical reactions. Part XXI. Simple methods for the radical deoxygenation of primary alcohols.☆ | DHR Barton, P Blundell, J Dorchak, DO Jang… - Tetrahedron, 1991 - Elsevier. 4 November 1991,. Tetrahedron. Volume 47, Issue 43,: Pages 8969-8984.
- The invention of radical reactions. Part XXIV. Relative rates of acylation and radical deoxygenation of secondary alcohols.☆ | DHR Barton, J Dorchak, JC Jaszberenyi - Tetrahedron, 1992 - Elsevier. 4 September 1992,. Tetrahedron,. Volume 48, Issue 36: Pages 7435-7446.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pentafluorophenyl chlorothionoformate, 1 g | sc-228910 | 1 g | $38.00 |
