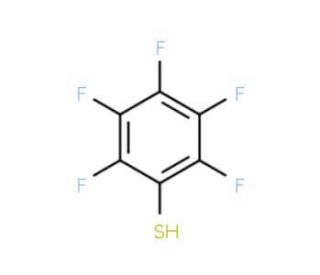
Pentafluorobenzenethiol (CAS 771-62-0)
See product citations (1)
QUICK LINKS
Pentafluorobenzenethiol (PFBT) is a highly fluorinated sulfur-containing molecule that exhibits a diverse range of applications in chemical synthesis, biochemistry, and materials science. Composed of five fluorine atoms, a benzene ring, and a thiol group, Pentafluorobenzenethiol serves as a valuable intermediate in the synthesis of various compounds, including pharmaceuticals, pesticides, and dyes. Its exceptional properties make it a promising candidate for biomedical applications, such as drug delivery systems and biosensors. In scientific research, Pentafluorobenzenethiol finds extensive use as an intermediate for synthesizing diverse compounds, including pharmaceuticals, pesticides, and dyes. It also contributes to the synthesis of fluorinated polymers and the production of fluorinated surfactants. Pentafluorobenzenethiol acts as a reagent in the synthesis of other fluorinated compounds like perfluorinated carboxylic acids and perfluorinated sulfonic acids. Furthermore, it acts as a catalyst in the synthesis of fluorinated organometallic compounds. Pentafluorobenzenethiol plays a significant role in the development of nanomaterials, including nanotubes and nanowires. Additionally, it serves as a reagent in synthesizing fluorinated biomolecules like peptides and proteins. The precise mechanism of action of Pentafluorobenzenethiol is not yet fully understood. However, it is believed that the fluorine atoms in Pentafluorobenzenethiol interact with the sulfur atom in the thiol group, forming a robust covalent bond. This bond contributes to Pentafluorobenzenethiol′s unique properties, such as high solubility in organic solvents and resistance to hydrolysis. Additionally, the fluorine atoms may interact with other molecules, such as proteins, forming strong non-covalent bonds that are responsible for Pentafluorobenzenethiol′s distinctive biological activity.
Pentafluorobenzenethiol (CAS 771-62-0) References
- Mild and general conditions for the cross-coupling of aryl halides with pentafluorobenzene and other perfluoroaromatics. | Lafrance, M., et al. 2006. Org Lett. 8: 5097-100. PMID: 17048852
- Gold nanoparticles with perfluorothiolate ligands. | Dass, A., et al. 2008. Langmuir. 24: 310-5. PMID: 18052299
- Formation and superlattice of long-range-ordered self-assembled monolayers of pentafluorobenzenethiols on Au(111). | Kang, H., et al. 2010. Langmuir. 26: 2983-5. PMID: 20131891
- Two-dimensional ordering of pentafluorobenzenethiol self-assembled monolayers on Au(111) prepared by ambient-pressure vapor deposition. | Kang, H., et al. 2010. Ultramicroscopy. 110: 666-9. PMID: 20189719
- Catalytic hydrodefluorination of pentafluorobenzene by [Ru(NHC)(PPh3)2(CO)H2]: a nucleophilic attack by a metal-bound hydride ligand explains an unusual ortho-regioselectivity. | Panetier, JA., et al. 2011. Angew Chem Int Ed Engl. 50: 2783-6. PMID: 21387487
- Formation of highly ordered and orientated gold islands: effect of immersion time on the molecular adlayer structure of pentafluorobenzenethiols (PFBT) SAMs on Au(111). | Azzam, W., et al. 2012. Langmuir. 28: 10192-208. PMID: 22690878
- Thiols as interfacial modifiers to enhance the performance and stability of perovskite solar cells. | Cao, J., et al. 2015. Nanoscale. 7: 9443-7. PMID: 25964159
- A Novel Alkylated Indacenodithieno[3,2-b]thiophene-Based Polymer for High-Performance Field-Effect Transistors. | Zhang, W., et al. 2016. Adv Mater. 28: 3922-7. PMID: 26514111
- Long-lived coherence in pentafluorobenzene as a probe of ππ(*) - πσ(*) vibronic coupling. | Hüter, O., et al. 2016. J Chem Phys. 145: 014302. PMID: 27394103
- Wettability Control of Gold Surfaces Modified with Benzenethiol Derivatives: Water Contact Angle and Thermal Stability. | Tatara, S., et al. 2016. J Nanosci Nanotechnol. 16: 3295-300. PMID: 27451620
- Hydrogen bonding vs. molecule-surface interactions in 2D self-assembly of [C60]fullerenecarboxylic acids. | Mezour, MA., et al. 2016. Nanoscale. 8: 16955-16962. PMID: 27714083
- On the Role of Contact Resistance and Electrode Modification in Organic Electrochemical Transistors. | Paterson, AF., et al. 2019. Adv Mater. 31: e1902291. PMID: 31343087
- Directional Doping and Cocrystallizing an Open-Shell Ag39 Superatom via Precursor Engineering. | Ma, XH., et al. 2022. ACS Nano. 16: 5507-5514. PMID: 35353504
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pentafluorobenzenethiol, 25 g | sc-263979 | 25 g | $275.00 | |||
Pentafluorobenzenethiol, 100 g | sc-263979A | 100 g | $798.00 |
