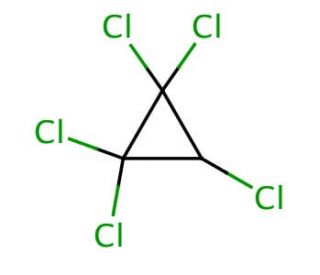
Molecular structure of Pentachlorocyclopropane, CAS Number: 6262-51-7
Pentachlorocyclopropane (CAS 6262-51-7)
Application:
Pentachlorocyclopropane is used to prepare tetrachlorocyclopropene via reaction with 18M aqueous KOH at 80-85°C and substituted phenyltrichlorocyclopropene derivatives
CAS Number:
6262-51-7
Purity:
≥88%
Molecular Weight:
214.31
Molecular Formula:
C3HCl5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Pentachlorocyclopropane is used to prepare tetrachlorocyclopropene via reaction with 18M aqueous KOH at 80-85°C and substituted phenyltrichlorocyclopropene derivatives. Pentachlorocyclopropane (PCCP) is a halogenated hydrocarbon and a persistent organic pollutant (POP). It is a volatile organic compound (VOC) with no color or odor. PCCP is derived from the chlorination of cyclopropane, a three-carbon ring structure with one double bond. Due to its widespread usage and subsequent release into the environment, PCCP is found as an environmental contaminant.
Pentachlorocyclopropane (CAS 6262-51-7) References
- Pentachlorocyclopropane/base complexes: matrix isolation infrared spectroscopic and density functional study of C-H- - -N hydrogen bonds. | Baker, AB., et al. 2005. J Phys Chem A. 109: 8280-9. PMID: 16834216
- Matrix and polymer soft-landing isolation of selected acids with pyridine and poly(4-vinylpyridine): a comparative infrared spectroscopic study of hydrogen bonding. | Lifschitz, AM., et al. 2012. J Phys Chem B. 116: 211-20. PMID: 22111986
- The evolution of cyclopropenium ions into functional polyelectrolytes. | Jiang, Y., et al. 2015. Nat Commun. 6: 5950. PMID: 25575214
- Phase-transfer and other types of catalysis with cyclopropenium ions. | Bandar, JS., et al. 2015. Chemistry. 21: 7365-8. PMID: 25820636
- Structure-Activity Relationship Studies of Cyclopropenimines as Enantioselective Brønsted Base Catalysts. | Bandar, JS., et al. 2015. Chem Sci. 6: 1537-1547. PMID: 26504512
- Macrosteres: The Deltic Guanidinium Ion. | Mishiro, K., et al. 2016. European J Org Chem. 2016: 1655-1659. PMID: 27790071
- Fluoride Ionic Liquids in Salts of Ethylmethylimidazolium and Substituted Cyclopropenium Cation Families. | Curnow, OJ., et al. 2018. Front Chem. 6: 603. PMID: 30619813
- Influence of Substituent Chain Branching on the Transfection Efficacy of Cyclopropenium-Based Polymers. | Brucks, SD., et al. 2017. Polymers (Basel). 9: PMID: 30970759
- Bis-aminocyclopropenylidene carbene borane catalyzed imine hydrogenation. | Huchenski, BSN., et al. 2019. Org Biomol Chem. 17: 6158-6164. PMID: 31183491
- Electrophotocatalysis with a Trisaminocyclopropenium Radical Dication. | Huang, H., et al. 2019. Angew Chem Int Ed Engl. 58: 13318-13322. PMID: 31306561
- Cyclopropenium Cationic-Based Covalent Organic Polymer-Enhanced Poly(ethylene oxide) Composite Polymer Electrolyte for All-Solid-State Li-S Battery. | Wang, Y., et al. 2021. ACS Appl Mater Interfaces. 13: 16469-16477. PMID: 33813826
- Cationic Cyclopropenium-Based Hyper-Crosslinked Polymer Enhanced Polyethylene Oxide Composite Electrolyte for All-Solid-State Li-S Battery. | Lian, S., et al. 2021. Nanomaterials (Basel). 11: PMID: 34685002
- Highly-fluorinated Triaminocyclopropenium Ionic Liquids. | Curnow, OJ. and Senthooran, R. 2022. Chem Asian J. 17: e202200139. PMID: 35239986
- Synthesis and physical properties of tris(dialkylamino)cyclopropenium dicyanamide ionic liquids. | Curnow, OJ., et al. 2018. RSC Adv. 8: 28313-28322. PMID: 35542454
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pentachlorocyclopropane, 5 g | sc-236308 | 5 g | $140.00 |
