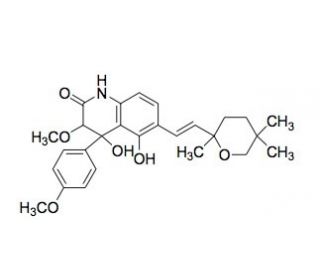
Penigequinolone A (CAS 180045-91-4)
QUICK LINKS
Penigequinolone A, a novel quinolone compound isolated from the fungal species Penicillium, is primarily studied for its unique biochemical properties and potential as a research tool in cellular and molecular biology. This compound is particularly noted for its ability to interfere with bacterial DNA gyrase, an enzyme critical for DNA replication and transcription. By inhibiting this enzyme, Penigequinolone A provides a valuable means to study bacterial growth mechanisms, DNA replication processes, and cellular responses to DNA stress. Additionally, the compound′s structure and function are explored for insights into the natural product biosynthesis pathways in fungi, shedding light on the genetic and enzymatic mechanisms underlying the production of secondary metabolites with bioactive properties. Research involving Penigequinolone A also extends to studies on quorum sensing and microbial communication, as its production by fungi suggests roles in microbial ecology and interspecies interactions. These applications make Penigequinolone A an important chemical for advancing our understanding of both microbial physiology and the potential for developing novel research tools based on natural compounds, thereby contributing to the fields of microbial ecology, genetics, and biochemistry.
Penigequinolone A (CAS 180045-91-4) References
- Phylogeny of Penicillium and the segregation of Trichocomaceae into three families. | Houbraken, J. and Samson, RA. 2011. Stud Mycol. 70: 1-51. PMID: 22308045
- Tandem prenyltransferases catalyze isoprenoid elongation and complexity generation in biosynthesis of quinolone alkaloids. | Zou, Y., et al. 2015. J Am Chem Soc. 137: 4980-3. PMID: 25859931
- Enzyme-catalyzed cationic epoxide rearrangements in quinolone alkaloid biosynthesis. | Zou, Y., et al. 2017. Nat Chem Biol. 13: 325-332. PMID: 28114276
- Copper-catalyzed trifluoromethylazidation and rearrangement of aniline-linked 1,7-enynes: access to CF3-substituted azaspirocyclic dihydroquinolin-2-ones and furoindolines. | Yu, LZ., et al. 2017. Chem Commun (Camb). 53: 8980-8983. PMID: 28744528
- Five New Records of the Family Aspergillaceae in Korea, Aspergillus europaeus, A. pragensis, A. tennesseensis, Penicillium fluviserpens, and P. scabrosum. | Nguyen, TTT., et al. 2020. Mycobiology. 48: 81-94. PMID: 32363036
- Natural 3,4-dihydro-2(1h)-quinolinones- Part II: animal, bacterial, and fungal sources. | Khadem, S. and Marles, RJ. 2024. Nat Prod Res. 1-14. PMID: 38564663
- Consistent production of penigequinolone A and B by Penicillium scabrosum | Larsen, T. O.Smedsgaard, J., et al. 1999. Biochemical systematics and ecology. 27(3): 329-332.
- New metabolites from Penicillium thymicola IBT 5891 | Ma, L. F., et al. 2017. Journal of Chemical Research. 41(2): 95-97.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Penigequinolone A, 500 µg | sc-202277 | 500 µg | $247.00 |
