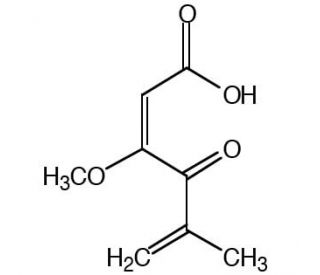
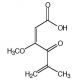
Penicillic acid (CAS 90-65-3)
See product citations (5)
QUICK LINKS
Penicillic acid is an organic compound that is extensively researched due to its role as a mycotoxin produced by various species of Penicillium and Aspergillus. It is a subject of interest in food safety and agricultural research to understand its occurrence in foodstuffs and feeds, as well as the conditions that favor its production. Studies on penicillic acid also involve its biosynthesis pathways and the genetic regulation of these pathways in fungi. In the field of toxin research, penicillic acid is used to investigate its mechanism of action, which includes the inhibition of certain enzymes and interference with macromolecular synthesis. Additionally, it is studied for its impact on microbial ecology, particularly how it affects interspecies interactions within microbial communities.
Penicillic acid (CAS 90-65-3) References
- Vicinal-thiol-containing molecules enhance but mono-thiol-containing molecules reduce nickel-induced DNA strand breaks. | Lynn, S., et al. 1999. Toxicol Appl Pharmacol. 160: 198-205. PMID: 10527919
- Inactivation of GDP-mannose dehydrogenase from Pseudomonas aeruginosa by penicillic acid identifies a critical active site loop. | Kimmel, JL. and Tipton, PA. 2005. Arch Biochem Biophys. 441: 132-40. PMID: 16111644
- Production of penicillic acid by Aspergillus sclerotiorum CGF. | Kang, SW., et al. 2007. Bioresour Technol. 98: 191-7. PMID: 16458001
- Compounds related to penicillic acid; synthesis of penicillic acid. | RAPHAEL, RA. 1948. J Chem Soc. 37: 1508-12. PMID: 18121538
- Tautomerism of penicillic acid. | MUNDAY, CW. 1949. Nature. 163: 443. PMID: 18224892
- Penicillic acid production in submerged culture. | Lindenfelser, LA. and Ciegler, A. 1977. Appl Environ Microbiol. 34: 553-6. PMID: 22310
- Determination of mycophenolic acid, penicillic acid, patulin, sterigmatocystin, and aflatoxins in cheese. | Siriwardana, MG. and Lafont, P. 1979. J Dairy Sci. 62: 1145-8. PMID: 229137
- Toxicity of penicillic acid for rat alveolar macrophages in vitro. | Sorenson, WG. and Simpson, J. 1986. Environ Res. 41: 505-13. PMID: 2430790
- Penicillic acid in fruits: method development, validation by liquid chromatography-tandem mass spectrometry and survey in southern China. | Guan, W., et al. 2021. J Sci Food Agric. 101: 2779-2787. PMID: 33140404
- Isolation and agricultural potential of penicillic acid against citrus canker. | Vieira, G., et al. 2022. J Appl Microbiol. 132: 3081-3088. PMID: 34927315
- Mutagenicity and inducibility of DNA single-strand breaks and chromosome aberrations by various mycotoxins. | Umeda, M., et al. 1977. Gan. 68: 619-25. PMID: 563356
- Acute toxicity of penicillic acid and rubratoxin B in dogs. | Hayes, AW., et al. 1977. Ann Nutr Aliment. 31: 711-21. PMID: 580698
- DNA-attacking ability of carcinogenic mycotoxins in recombination-deficient mutant cells of Bacillus subtilis. | Ueno, Y. and Kubota, K. 1976. Cancer Res. 36: 445-51. PMID: 816461
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Penicillic acid, 5 mg | sc-205796 | 5 mg | $130.00 | |||
Penicillic acid, 25 mg | sc-205796A | 25 mg | $281.00 |
