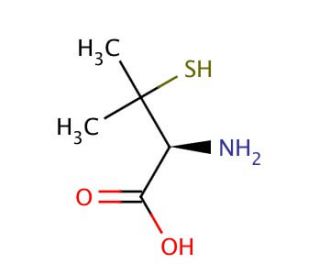
Penicillamine (CAS 52-67-5)
QUICK LINKS
Penicillamine is a compound with a variety of applications in research, particularly in the field of chemistry and biochemistry. In studies of metal chelation, penicillamine is used due to its ability to bind divalent metal ions, which is beneficial for understanding the chelating properties of thiol-containing ligands. Its role in the synthesis of complex metallo-organic structures is also a focus, as these structures may have interesting catalytic or structural properties. In protein chemistry, researchers utilize penicillamine to induce and study the formation of disulfide bonds, which are critical for the stability and function of many proteins. Through these applications, penicillamine contributes to a deeper understanding of metal ion interactions with organic molecules and the folding and stability of polypeptides. Additionally, the compound′s enantiomeric forms are of interest in the study of chiral resolution techniques and the synthesis of asymmetric catalysts.
Penicillamine (CAS 52-67-5) References
- Nitric oxide donors regulate nitric oxide synthase in bovine pulmonary artery endothelium. | Chen, JX., et al. 2001. J Cell Physiol. 186: 116-23. PMID: 11147806
- Suppression of urease levels in Streptococcus salivarius by cysteine, related compounds and by sulfide. | Sissons, CH. and Yakub, S. 2000. Oral Microbiol Immunol. 15: 317-24. PMID: 11154424
- Application of an unusual ninhydrin-based reaction for the indirect chiral resolution of D,L-penicillamine. | Sotgia, S., et al. 2011. Talanta. 85: 1783-5. PMID: 21872019
- Orthogonal Cysteine-Penicillamine Disulfide Pairing for Directing the Oxidative Folding of Peptides. | Zheng, Y., et al. 2015. J Am Chem Soc. 137: 15094-7. PMID: 26588670
- Theoretical spectroscopic insights of tautomers and enantiomers of penicillamine. | Mendes, J., et al. 2017. Spectrochim Acta A Mol Biomol Spectrosc. 184: 308-317. PMID: 28525866
- Liquid chromatography/tandem mass spectrometric analysis of penicillamine for its pharmacokinetic evaluation in dogs. | Lehner, AF., et al. 2020. Toxicol Mech Methods. 30: 687-702. PMID: 32854553
- Inspecting the structural characteristics of chiral drug penicillamine under different pH conditions using Raman optical activity spectroscopy and DFT calculations. | Guo, YT., et al. 2021. Phys Chem Chem Phys. 23: 22119-22132. PMID: 34580687
- Cytogenetic studies in patients treated with penicillamine. | Jensen, MK., et al. 1979. Mutat Res. 67: 357-9. PMID: 384237
- Penicillamine nephropathy and iron. | Harkness, JA. and Blake, DR. 1982. Lancet. 2: 1368-9. PMID: 6129465
- Penicillamine in rheumatoid disease. | Thrift, EG. and Lewis, B. 1983. Aust Fam Physician. 12: 870-1. PMID: 6670953
- The side effects of penicillamine therapy in rheumatoid arthritis. | Huskisson, EC. 1981. J Rheumatol Suppl. 7: 146-8. PMID: 6939876
- Penicillamine-induced polymyositis-dermatomyositis. | Petersen, J., et al. 1978. Scand J Rheumatol. 7: 113-7. PMID: 705267
- Pemphigus in rheumatoid arthritis treated with penicillamine. | Livden, JK., et al. 1981. Scand J Rheumatol. 10: 95-6. PMID: 7244584
- Penicillamine kinetics in normal subjects. | Bergstrom, RF., et al. 1981. Clin Pharmacol Ther. 30: 404-13. PMID: 7273605
- The pharmacokinetics of penicillamine in a female mongrel dog. | Bergstrom, RF., et al. 1981. J Pharmacokinet Biopharm. 9: 603-21. PMID: 7334461
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Penicillamine, 1 g | sc-205795 | 1 g | $46.00 | |||
Penicillamine, 5 g | sc-205795A | 5 g | $96.00 |
