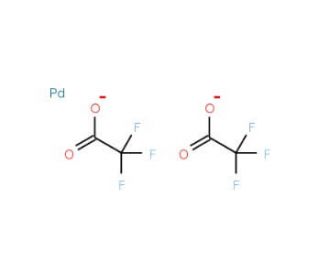
Palladium(II) trifluoroacetate (CAS 42196-31-6)
QUICK LINKS
Palladium(II) trifluoroacetate is a chemical of significant interest in the field of synthetic organic chemistry, particularly as a catalyst in various carbon-carbon bond-forming reactions such as the Suzuki coupling, which is pivotal in constructing complex organic molecules. Its efficiency and selectivity make it useful in the synthesis of fine chemicals and in the field of catalysis research. Researchers analyze into its mechanisms of action, exploring the intricacies of oxidative addition and reductive elimination processes that are central to its catalytic activity. In materials science, this compound is investigated for its potential role in the fabrication of novel materials through controlled organic reactions. Investigations into its catalytic properties extend to its use in the field of green chemistry, where it helps to promote more sustainable chemical processes.
Palladium(II) trifluoroacetate (CAS 42196-31-6) References
- Pd-catalyzed asymmetric hydrogenation of alpha-fluorinated iminoesters in fluorinated alcohol: a new and catalytic enantioselective synthesis of fluoro alpha-amino acid derivatives. | Abe, H., et al. 2001. Org Lett. 3: 313-5. PMID: 11428002
- The reactions of palladium(II), thallium(III) and lead(IV) trifluoroacetates with 3beta-acetoxyandrost-5-en-17-one: crystal structure of the first trifluoroacetate bridged 5,6,7-pi-allyl steroid palladium dimer. | Ruddock, PL., et al. 2004. Steroids. 69: 193-9. PMID: 15072921
- On the mechanism of the palladium(II)-catalyzed decarboxylative olefination of arene carboxylic acids. Crystallographic characterization of non-phosphine palladium(II) intermediates and observation of their stepwise transformation in Heck-like processes. | Tanaka, D., et al. 2005. J Am Chem Soc. 127: 10323-33. PMID: 16028944
- Mechanism and enantioselectivity in palladium-catalyzed conjugate addition of arylboronic acids to β-substituted cyclic enones: insights from computation and experiment. | Holder, JC., et al. 2013. J Am Chem Soc. 135: 14996-5007. PMID: 24028424
- Palladium Catalyzed C-Arylation of Amino Acid Derived Hydantoins. | Fernández-Nieto, F., et al. 2015. Org Lett. 17: 3838-41. PMID: 26200165
- From Pincer to Paddlewheel: C-H and C-S Bond Activation at Bis(2-pyridylthio)methane by Palladium(II). | Halder, P., et al. 2019. Inorg Chem. 58: 2270-2274. PMID: 30698431
- Ligand Rearrangement Leads to Tetrahydrothiophene-Functionalized N,S-Heterocyclic Carbene Palladium(II) Complexes. | Romine, AM., et al. 2021. Organometallics. 40: 2311-2319. PMID: 34433997
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Palladium(II) trifluoroacetate, 1 g | sc-253234 | 1 g | $153.00 |
