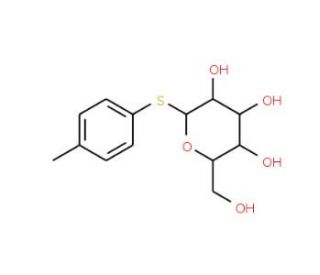
p-Tolyl 1-thio-β-D-glucopyranoside (CAS 1152-39-2)
QUICK LINKS
p-Tolyl 1-thio-β-D-glucopyranoside is a thioglycoside extensively utilized in glycoscience research, particularly for its role as a glycosyl donor in synthetic carbohydrate chemistry. This compound is characterized by its sulfur atom, which replaces the oxygen atom in the glycosidic bond, enhancing its stability against hydrolysis and making it a robust intermediate in glycosylation reactions. The p-tolyl group provides additional stability and facilitates the activation of the glycosyl donor in the presence of promoters such as N-iodosuccinimide (NIS) and trifluoromethanesulfonic acid (TfOH). In research, p-tolyl 1-thio-β-D-glucopyranoside is employed to study the mechanisms of glycosylation, focusing on the formation and selectivity of glycosidic bonds. Its application is crucial in the synthesis of complex oligosaccharides and glycoconjugates, which are important for exploring carbohydrate-protein interactions and cell surface recognition. This compound is also used in the development of glycomimetics, which are synthetic analogs of naturally occurring glycans, to investigate their role in biological processes. Additionally, p-tolyl 1-thio-β-D-glucopyranoside is utilized in enzymatic studies to explore the specificity and catalytic mechanisms of glycosyltransferases and glycosidases. Its versatility and stability make it a valuable tool in advancing the understanding of carbohydrate chemistry and glycosylation pathways.
p-Tolyl 1-thio-β-D-glucopyranoside (CAS 1152-39-2) References
- Selective electrochemical glycosylation by reactivity tuning. | France, RR., et al. 2004. Org Biomol Chem. 2: 2195-202. PMID: 15280955
- Highly efficient syntheses of hyaluronic acid oligosaccharides. | Huang, L. and Huang, X. 2007. Chemistry. 13: 529-40. PMID: 17039562
- Synthesis of Defined and Functionalized Glycans of Lipoteichoic Acid: A Cell Surface Polysaccharide from Clostridium difficile. | Yu, K., et al. 2017. Org Lett. 19: 3123-3126. PMID: 28548838
- Chemical synthesis of amphiphilic glycoconjugates: Access to amino, fluorinated and sulfhydryl oleyl glucosides. | Porter, J., et al. 2023. Carbohydr Res. 530: 108854. PMID: 37329646
- Reaction Rate and Stereoselectivity Enhancement in Glycosidations with O-Glycosyl Trihaloacetimidate Donors due to Catalysis by a Lewis Acid-Nitrile Cooperative Effect. | Li, T., et al. 2024. J Org Chem. 89: 7865-7876. PMID: 38805026
- Concise synthesis of two trisaccharides related to the saponin isolated from Centratherum anthelminticum. | Mandal, Santanu and Balaram Mukhopadhyay. 2007. Tetrahedron. 63.46: 11363-11370.
- Thio‐β‐D‐glucosides: Synthesis and Evaluation as Glycosidase Inhibitors and Activators. | Samoshin, Andrey V., et al. 2014. International journal of carbohydrate chemistry. 2014.1: 941059.
- Efficient one-pot per-O-acetylation–thioglycosidation of native sugars, 4, 6-O-arylidenation and one-pot 4, 6-O-benzylidenation–acetylation of S-/O-glycosides catalyzed by Mg (OTf) 2. | Mukherjee, Mana Mohan, et al. 2016. RSC advances. 6.111: 109301-109314.
- Alternative Routes to 4, 6‐O‐Benzylidene β‐Thioglycosides. | Imperio, Daniela, et al. 2024. Helvetica Chimica Acta. 107: e202300193.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
p-Tolyl 1-thio-β-D-glucopyranoside, 100 mg | sc-257958 | 100 mg | $54.00 |
