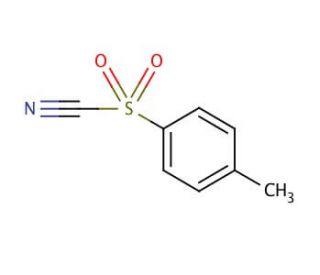
p-Toluenesulfonyl cyanide (CAS 19158-51-1)
QUICK LINKS
p-Toluenesulfonyl cyanide is a widely utilized, high toxicity, chemical compound in organic synthesis. It presents as a colorless, crystalline solid with a pungent odor and exhibits solubility in organic solvents. The applications of p-Toluenesulfonyl cyanide cyanide extend to various scientific research endeavors. It finds utility in peptide synthesis, the synthesis of heterocyclic compounds, and the creation of organometallic compounds. p-Toluenesulfonyl cyanide cyanide functions as an electrophilic reagent, possessing the ability to react with nucleophiles. The reaction entails the formation of a p-Toluenesulfonyl cyanideate anion upon the interaction with a nucleophile, which is subsequently attacked by the nucleophile. This sequence of events culminates in the formation of a novel C-C bond.
p-Toluenesulfonyl cyanide (CAS 19158-51-1) References
- Tin-free radical-mediated C[bond]C-bond formations with alkyl allyl sulfones as radical precursors. | Kim, S. and Lim, CJ. 2002. Angew Chem Int Ed Engl. 41: 3265-7. PMID: 12207411
- 2-azabicyclo[2.2.2]octa-3,5-dione via a nitrile Diels-Alder reaction. | McClure, CK. and Link, JS. 2003. J Org Chem. 68: 8256-7. PMID: 14535813
- Synthesis of 3-substituted bicyclic imidazo[1,2-d][1,2,4]thiadiazoles and tricyclic benzo[4,5]imidazo[1,2-d][1,2,4]thiadiazoles. | Leung-Toung, R., et al. 2005. J Org Chem. 70: 6230-41. PMID: 16050682
- Synthesis and characterization of 2,4-pentadiynenitrile--a key compound in space science. | Trolez, Y. and Guillemin, JC. 2005. Angew Chem Int Ed Engl. 44: 7224-6. PMID: 16224748
- A click chemistry approach to tetrazoles by Huisgen 1,3-dipolar cycloaddition: synthesis of 5-sulfonyl tetrazoles from azides and sulfonyl cyanides. | Demko, ZP. and Sharpless, KB. 2002. Angew Chem Int Ed Engl. 41: 2110-3. PMID: 19746612
- A click chemistry approach to tetrazoles by Huisgen 1,3-dipolar cycloaddition: synthesis of 5-acyltetrazoles from azides and acyl cyanides. | Demko, ZP. and Sharpless, KB. 2002. Angew Chem Int Ed Engl. 41: 2113-6. PMID: 19746613
- Electrophilic Cyanation of Boron Enolates: Efficient Access to Various β-Ketonitrile Derivatives. | Kiyokawa, K., et al. 2016. Angew Chem Int Ed Engl. 55: 10458-62. PMID: 27417187
- Enantioselective Electrophilic Cyanation of Boron Enolates: Scope and Mechanistic Studies. | Nagata, T., et al. 2018. Chemistry. 24: 17027-17032. PMID: 30256466
- Stereospecific Oxycyanation of Alkenes with Sulfonyl Cyanide. | Kiyokawa, K., et al. 2023. Angew Chem Int Ed Engl. 62: e202218743. PMID: 36702752
- Reactions of p-toluenesulfonyl chloride and p-toluenesulfonyl cyanide with sodium cyanide and with sodium p-toluenesulfinate | Pews, R. G., & Corson, F. P. 1971. The Journal of Organic Chemistry. 36(12): 1654-1659.
- O-Sulfinylation of alcohols with methanesulfonyl cyanide or p-toluenesulfonyl cyanide | Barton, D. H., Jaszberenyi, J. C., & Theodorakis, E. A. 1991. Tetrahedron. 47(44): 9167-9178.
- Preparation of polyfunctional nitriles by the cyanation of functionalized organozinc halides with p-toluenesulfonyl cyanide | Klement, I., Lennick, K., Tucker, C. E., & Knochel, P. 1993. Tetrahedron letters. 34(29): 4623-4626.
- Rh (III)‐Catalyzed Direct C− H Cyanation of Arenes with p‐Toluenesulfonyl Cyanide | Liu, M., You, E., Cao, W., & Shi, J. 2019. Asian Journal of Organic Chemistry. 8(10): 1850-1853.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
p-Toluenesulfonyl cyanide, 1 g | sc-250632 | 1 g | $60.00 |
