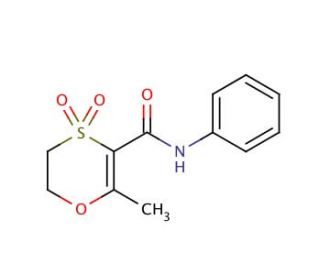
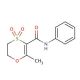
Molecular structure of Oxycarboxine, CAS Number: 5259-88-1
Oxycarboxine (CAS 5259-88-1)
CAS Number:
5259-88-1
Molecular Weight:
267.30
Molecular Formula:
C12H13NO4S
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Oxycarboxine is derived from carboxine, a employed carboxylic acid in chemical synthesis. Within scientific research, oxycarboxine serves diverse purposes. It acts as a reagent in organic synthesis, facilitating the formation of carboxylic acids, esters, and amides. Furthermore, it plays a role in peptide and protein synthesis. In organic synthesis, oxycarboxine functions as a nucleophile, readily forming chemical bonds with other compounds. Its high reactivity endows it with strong nucleophilic properties, allowing rapid bond formation. Additionally, oxycarboxine exhibits the ability to form hydrogen bonds with other compounds, enabling the stabilization of reaction products.
Oxycarboxine (CAS 5259-88-1) References
- Effect of the systemic fungicide carboxin on electron transport function in membranes of Micrococcus denitrificans. | Tucker, AN. and Lillich, TT. 1974. Antimicrob Agents Chemother. 6: 572-8. PMID: 15825307
- Application of ultrahigh-performance liquid chromatography and electrospray ionization quadrupole orbitrap high-resolution mass spectrometry for determination of 166 pesticides in fruits and vegetables. | Wang, J., et al. 2012. J Agric Food Chem. 60: 12088-104. PMID: 23163727
- Carboxin and its major metabolites residues in peanuts: Levels, dietary intake and chronic intake risk assessment. | Wei, D., et al. 2019. Food Chem. 275: 169-175. PMID: 30724184
- Biomonitoring of pesticides, pharmaceuticals and illicit drugs in a freshwater invertebrate to estimate toxic or effect pressure. | Miller, TH., et al. 2019. Environ Int. 129: 595-606. PMID: 31053240
- Efficacy of Fungicides Applied for Protectant and Curative Activity Against Myrtle Rust. | Pathan, AK., et al. 2020. Plant Dis. 104: 2123-2129. PMID: 32539594
- Multiresidues of environmental contaminants in bats from Turkey. | Kuzukiran, O., et al. 2021. Chemosphere. 282: 131022. PMID: 34090000
- Knowledge integration and decision support for accelerated discovery of antibiotic resistance genes. | Youn, J., et al. 2022. Nat Commun. 13: 2360. PMID: 35487919
- Pesticides: nomenclature, specification, analysis, use, and residues in foods. | Lowe, DA. and Stiles, AR. 1973. Bull World Health Organ. 49: 169-204. PMID: 4545324
- Silver nitrate-bromophenol blue and potassium permanganate-sulphuric acid as chromogenic reagents for the detection of carboxin and oxycarboxin in thin-layer chromatography. | Tripathi, RK. and Bhaktavatsalam, G. 1973. J Chromatogr. 87: 282-6. PMID: 4772377
- Mode of action of oxathiin systemic fungicides. I. Effect of carboxin and oxycarboxin on the general metabolism of several basidiomycetes. | Mathre, DE. 1970. Phytopathology. 60: 671-6. PMID: 5441309
- The rate of penetration of leaf tissues by the systemic fungicides benodanil (2-iodobenzanilide) and oxycarboxin (2,3-dihydro-6 methyl-5 phenylcarbamoyl-1,4-oxathin-4,4-dioxide). | Udeogalanya, AC. 1984. Beitr Trop Landwirtsch Veterinarmed. 22: 91-4. PMID: 6732737
- Degradation of carboxin (Vitavax) and oxycarboxin (Plantvax) by Pseudomonas aeruginosa isolated from soil. | Balasubramanya, RH., et al. 1980. J Environ Sci Health B. 15: 485-505. PMID: 6776177
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Oxycarboxine, 100 mg | sc-250622 | 100 mg | $99.00 |
