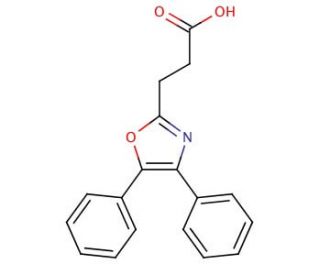
Molecular structure of Oxaprozin, CAS Number: 21256-18-8
Oxaprozin (CAS 21256-18-8)
Alternate Names:
4,5-Diphenyl-2-oxazolepropanoic acid
Application:
Oxaprozin is a non-narcotic anti-inflammatory agent shown to inhibit prostaglandin synthesis
CAS Number:
21256-18-8
Molecular Weight:
293.32
Molecular Formula:
C18H15NO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Oxaprozin undergoes conversion to its active form, oxaprozin acid. Once formed, oxaprozin acid acts by binding to the cyclooxygenase (COX) enzymez, effectively impeding their activity. These enzymes are for the conversion of arachidonic acid to prostaglandins and thromboxanes. Consequently, the synthesis of prostaglandins, hormones that play a role in the inflammatory response, is diminished. Additionally, oxaprozin inhibits the production of other inflammatory mediators, including leukotrienes and cytokines. By exerting these effects, oxaprozin helps to mitigate inflammation.
Oxaprozin (CAS 21256-18-8) References
- Oxaprozin-induced symptomatic hepatotoxicity. | Kethu, SR., et al. 1999. Ann Pharmacother. 33: 942-4. PMID: 10492496
- A review of the emerging profile of the anti-inflammatory drug oxaprozin. | Dallegri, F., et al. 2005. Expert Opin Pharmacother. 6: 777-85. PMID: 15934904
- Oxaprozin: a new NSAID. | al-Faks, MA. and Pugh, MC. 1992. Orthop Rev. 21: 558, 560-3. PMID: 1603605
- Synthesis, characterization and antiproliferative activity of transition metal complexes with 3-(4,5-diphenyl-1,3-oxazol-2-yl)propanoic acid (oxaprozin). | Božić, BD., et al. 2012. Chem Pharm Bull (Tokyo). 60: 865-9. PMID: 22790819
- Oxaprozin-Loaded Lipid Nanoparticles towards Overcoming NSAIDs Side-Effects. | Lopes-de-Araújo, J., et al. 2016. Pharm Res. 33: 301-14. PMID: 26350105
- Oxaprozin: A new hope in the modulation of matrix metalloproteinase 9 activity. | Ianni, A., et al. 2019. Chem Biol Drug Des. 93: 811-817. PMID: 30582279
- The study of ultrasound and iontophoresis on oxaprozin transdermal penetration using surface-enhanced Raman spectroscopy. | Liu, S., et al. 2020. Drug Deliv Transl Res. 10: 83-92. PMID: 31407271
- Oxaprozin Analogues as Selective RXR Agonists with Superior Properties and Pharmacokinetics. | Schierle, S., et al. 2021. J Med Chem. 64: 5123-5136. PMID: 33793232
- Oxaprozin. A preliminary review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy. | Todd, PA. and Brogden, RN. 1986. Drugs. 32: 291-312. PMID: 3536423
- Anticonvulsant activity of oxaprozin in a rat model of pentylenetetrazole-induced seizure by targeting oxidative stress and SIRT1/PGC1α signaling. | Khatami, P., et al. 2022. Can J Physiol Pharmacol. 100: 534-541. PMID: 35395161
- Application of CO2 Supercritical Fluid to Optimize the Solubility of Oxaprozin: Development of Novel Machine Learning Predictive Models. | Alshahrani, SM., et al. 2022. Molecules. 27: PMID: 36144490
- Two Binding Sites of SARS-CoV-2 Macrodomain 3 Probed by Oxaprozin and Meclomen. | Li, J., et al. 2022. J Med Chem. 65: 15227-15237. PMID: 36356292
- Oxaprozin pharmacokinetics in the elderly. | Greenblatt, DJ., et al. 1985. Br J Clin Pharmacol. 19: 373-8. PMID: 3986088
Inhibitor of:
C6, CatSperβ, PGT, PPARα, and PTG.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Oxaprozin, 5 mg | sc-215641 | 5 mg | $94.00 | |||
Oxaprozin, 10 mg | sc-215641B | 10 mg | $181.00 | |||
Oxaprozin, 50 mg | sc-215641A | 50 mg | $218.00 | |||
Oxaprozin, 100 mg | sc-215641C | 100 mg | $281.00 | |||
Oxaprozin, 1 g | sc-215641D | 1 g | $302.00 | |||
Oxaprozin, 2.5 g | sc-215641E | 2.5 g | $396.00 |
