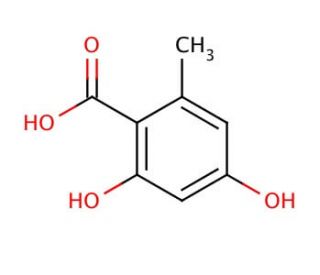
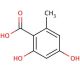
Orsellinic acid (CAS 480-64-8)
See product citations (1)
QUICK LINKS
Orsellinic acid is an intriguing organic compound, classified as a phenolic acid, that plays a pivotal role in the realm of natural product chemistry and biosynthetic research. Its structure is characterized by a benzene ring substituted with hydroxyl groups, which is foundational in the synthesis of many complex fungal metabolites and lichens. Orsellinic acid serves as a building block in the biosynthesis of various secondary metabolites, including mycotoxins and pigments, through polyketide pathways. This process involves the iterative addition of acetyl-CoA units, catalyzed by polyketide synthase enzymes, demonstrating a fascinating aspect of natural product synthesis. Researchers are particularly interested in orsellinic acid due to its involvement in these biosynthetic pathways, offering insights into the molecular mechanisms underlying the diversity of natural compounds. Additionally, understanding the biosynthesis of orsellinic acid and its derivatives can inform the development of novel bioengineering approaches to produce these compounds in microbial or synthetic hosts, which is of great interest for applications in biotechnology and the chemical industry.
Orsellinic acid (CAS 480-64-8) References
- Studies in the biochemistry of micro-organisms. 110. Production and biosynthesis of orsellinic acid by Penicillium madriti G. Smith. | BIRKINSHAW, JH. and GOWLLAND, A. 1962. Biochem J. 84: 342-7. PMID: 13869400
- Globosumones A-C, cytotoxic orsellinic acid esters from the Sonoran desert endophytic fungus Chaetomium globosum. | Bashyal, BP., et al. 2005. J Nat Prod. 68: 724-8. PMID: 15921417
- Cytotoxic activity of orsellinates. | Gomes, AT., et al. 2006. Z Naturforsch C J Biosci. 61: 653-7. PMID: 17137109
- Platelet activating factor-induced neuronal apoptosis is initiated independently of its G-protein coupled PAF receptor and is inhibited by the benzoate orsellinic acid. | Ryan, SD., et al. 2007. J Neurochem. 103: 88-97. PMID: 17877634
- Plant phenolics regulate neoplastic cell growth and survival: a quantitative structure-activity and biochemical analysis. | Harris, CS., et al. 2007. Can J Physiol Pharmacol. 85: 1124-38. PMID: 18066115
- Radical-scavenging activity of orsellinates. | Lopes, TI., et al. 2008. Chem Pharm Bull (Tokyo). 56: 1551-4. PMID: 18981604
- VeA and MvlA repression of the cryptic orsellinic acid gene cluster in Aspergillus nidulans involves histone 3 acetylation. | Bok, JW., et al. 2013. Mol Microbiol. 89: 963-74. PMID: 23841751
- Fusarium graminearum PKS14 is involved in orsellinic acid and orcinol synthesis. | Jørgensen, SH., et al. 2014. Fungal Genet Biol. 70: 24-31. PMID: 25011010
- A Novel Class of Plant Type III Polyketide Synthase Involved in Orsellinic Acid Biosynthesis from Rhododendron dauricum. | Taura, F., et al. 2016. Front Plant Sci. 7: 1452. PMID: 27729920
- Biochemical and genetic basis of orsellinic acid biosynthesis and prenylation in a stereaceous basidiomycete. | Braesel, J., et al. 2017. Fungal Genet Biol. 98: 12-19. PMID: 27903443
- Identification of the orsellinic acid synthase PKS63787 for the biosynthesis of antroquinonols in Antrodia cinnamomea. | Yu, PW., et al. 2017. Appl Microbiol Biotechnol. 101: 4701-4711. PMID: 28255687
- Enzymology and biosynthesis of the orsellinic acid derived medicinal meroterpenoids. | Tao, H. and Abe, I. 2021. Curr Opin Biotechnol. 69: 52-59. PMID: 33383296
- Orsellinic acid-loaded chitosan nanoparticles in gelatin/nanohydroxyapatite scaffolds for bone formation in vitro. | Yadav, LR., et al. 2022. Life Sci. 299: 120559. PMID: 35447131
- Studies on the biosynthesis of phenols in fungi. Conversion of [14C]orsellinic acid and [14C]orcinol into fumigatol by Aspergillus fumigatus I.M.I. 89353. | Packter, NM. 1966. Biochem J. 98: 353-9. PMID: 5296209
- Isolation and identification of orsellinic acid and penicillic acid produced by Penicillium fennelliae Stolk. | Van Eijk, GW. 1969. Antonie Van Leeuwenhoek. 35: 497-504. PMID: 5311576
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Orsellinic acid, 10 mg | sc-202752 | 10 mg | $112.00 | |||
Orsellinic acid, 50 mg | sc-202752A | 50 mg | $459.00 | |||
Orsellinic acid, 250 mg | sc-202752B | 250 mg | $1224.00 |
