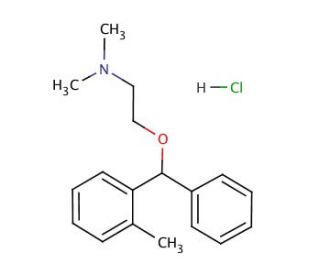
Orphenadrine hydrochloride (CAS 341-69-5)
QUICK LINKS
Orphenadrine hydrochloride is a compound that functions as a muscarinic antagonist, blocking the action of acetylcholine at muscarinic receptors in the central nervous system. Orphenadrine Hydrochloride inhibits the parasympathetic nervous system, leading to a decrease in smooth muscle contraction and glandular secretions. Orphenadrine hydrochloride also has a mild analgesic effect, which may be attributed to its ability to inhibit the release of substance P in the spinal cord. At the molecular level, Orphenadrine hydrochloride acts by binding to and blocking muscarinic acetylcholine receptors, thereby preventing the downstream signaling cascade that would normally occur upon acetylcholine binding. This interference with muscarinic receptor activity results in the suppression of parasympathetic nervous system function, which may be useful for studying the role of acetylcholine in various physiological processes.
Orphenadrine hydrochloride (CAS 341-69-5) References
- Orphenadrine poisoning in a child: clinical and analytical data. | Van Herreweghe, I., et al. 1999. Intensive Care Med. 25: 1134-6. PMID: 10551971
- Tetanus-like syndrome secondary to metoclopramide administration. | Mazza, A., et al. 2000. Ann Ital Med Int. 15: 301-2. PMID: 11202633
- Solid-state structure of orphenadrine hydrochloride and conformational comparisons with diphenhydramine hydrochloride and nefopam hydrochloride. | Glaser, R., et al. 1992. J Pharm Sci. 81: 858-62. PMID: 1432628
- [Trial of orphenadrine hydrochloride in the neurological sequels of brain diseases in children]. | MISES, R. and BRICOUT, J. 1960. Ann Med Psychol (Paris). 118(2): 155-60. PMID: 14422895
- Acetylcholine and choline levels in rabbit fetuses exposed to anticholinergics. | McBride, WG. and Hicks, LJ. 1987. Int J Dev Neurosci. 5: 117-25. PMID: 3503493
- Investigations of the Interaction Mechanism Between Orphenadrine Hydrochloride and Bovine Serum Albumin by Spectroscopic and Voltammetric Techniques. | Gokavi, NM., et al. 2023. J Fluoresc.. PMID: 36976401
- Orphenadrine serum levels in a poisoned patient. | Furlanut, M., et al. 1985. Hum Toxicol. 4: 331-3. PMID: 4007897
- Comparison of phenobarbital with orphenadrine hydrochloride and tofenacine hydrochloride for enzyme-inducing activity in young and adult rats. | Funcke, AB. and Timmerman, H. 1973. Arch Int Pharmacodyn Ther. 205: 213-7. PMID: 4766547
- The effect of orphenadrine hydrochloride on the acetylcholine concentration in rat brain. | Hespe, W., et al. 1969. Int J Neuropharmacol. 8: 471-4. PMID: 5344490
- Investigation into the metabolic fate of orphenadrine hydrochloride after oral administration to male rats. | Hespe, W., et al. 1965. Arch Int Pharmacodyn Ther. 156: 180-200. PMID: 5858833
- Difference between single and multiple dose pharmacokinetics of orphenadrine hydrochloride in man. | Labout, JJ., et al. 1982. Eur J Clin Pharmacol. 21: 343-50. PMID: 7056281
- The bioavailability of orphenadrine hydrochloride after intramuscular and oral administration. | Rutigliano, G. and Labout, JJ. 1982. J Int Med Res. 10: 447-50. PMID: 7152086
- Atropine substitutes and the writhing syndrome in mice. | Kelkar, MS. 1977. Arch Int Pharmacodyn Ther. 229: 133-7. PMID: 931459
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Orphenadrine hydrochloride, 5 g | sc-255405 | 5 g | $63.00 |
