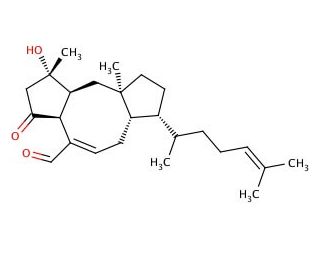
Molecular structure of Ophiobolin C, CAS Number: 19022-51-6
Ophiobolin C (CAS 19022-51-6)
Application:
Ophiobolin C is an inhibitor of human CCR5 binding to HIV-1 gp120
CAS Number:
19022-51-6
Purity:
≥95%
Molecular Weight:
386.6
Molecular Formula:
C25H38O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Ophiobolin C is a phytotoxic metabolite from the Ophiobolin class produced by many species of the genus Bipolaris. Studies suggest that Ophiobolin C inhibits CCR5 binding to the envelop protein gp120 and CD4, which is responsible for mediating the entry of HIV-1 into cells. In addition, the culture medium is a key component for the production of a specific Ophiobolin from the same organism. It has been reported that B. maydis will produce Ophiobolin A (sc-202266), B (sc-202267), and L if cultured in liquid medium, while in contrast produce Ophiobolin C and K in solid substratum medium. The structure of Ophiobolin C is comprised of five isoprene units linked together to form the backbone of the molecule.
Ophiobolin C (CAS 19022-51-6) References
- The biology of ophiobolins. | Au, TK., et al. 2000. Life Sci. 67: 733-42. PMID: 10968403
- Isolation and structure of antagonists of chemokine receptor (CCR5). | Jayasuriya, H., et al. 2004. J Nat Prod. 67: 1036-8. PMID: 15217290
- Phytotoxins from the pathogenic fungi Drechslera maydis and Drechslera sorghicola. | Sugawara, F., et al. 1987. Proc Natl Acad Sci U S A. 84: 3081-5. PMID: 16593832
- Strategies for the synthesis of fusicoccanes by Nazarov reactions of dolabelladienones: total synthesis of (+)-fusicoauritone. | Williams, DR., et al. 2007. Angew Chem Int Ed Engl. 46: 915-8. PMID: 17171749
- Cancer stem cell drugs target K-ras signaling in a stemness context. | Najumudeen, AK., et al. 2016. Oncogene. 35: 5248-5262. PMID: 26973241
- Multiple Oxidative Modifications in the Ophiobolin Biosynthesis: P450 Oxidations Found in Genome Mining. | Narita, K., et al. 2016. Org Lett. 18: 1980-3. PMID: 27116000
- The Biosynthesis and Transport of Ophiobolins in Aspergillus ustus 094102. | Yan, J., et al. 2022. Int J Mol Sci. 23: PMID: 35163826
- Ophiobolin M and analogues, noncompetitive inhibitors of ivermectin binding with nematocidal activity. | Tsipouras, A., et al. 1996. Bioorg Med Chem. 4: 531-6. PMID: 8735840
- The incredible fungal genus -Drechslera - and its phytotoxic ophiobolins | , et al. (1988). Phytoparasitica volume 16, pages 145–152 (1988). volume 16,: pages 145–152.
- Synthetic studies directed toward naturally occurring cyclooctanoids. 1. A total synthesis of (.+-.)-ceroplastol I. | Robert K. Boeckman Jr., Argyrios Arvanitis, and Matthew E. Voss. 1989,. J. Am. Chem. Soc. 111, 7,: 2737–2739.
- Variecolin, a sesterterpenoid of novel skeleton from Aspergillus variecolor MF138 | Otto D. Hensens, Debra Zink, Joanne M. Williamson, Victor J. Lotti, Raymond S. L. Chang, and Michael A. Goetz. 1991. J. Org. Chem. 1991, 56, 10, 3399–3403., 56, 10,: 3399–3403.
- Ni(II)/Cr(II)-Mediated Coupling Reaction: Beneficial Effects of 4-tert-Butylpyridine as an Additive and Development of New and Improved Workup Procedures | DP Stamos, XC Sheng, SS Chen, Y Kishi. September 1997,. Tetrahedron Letters. Volume 38, Issue 36, 8: Pages 6355-6358.
- Cytotoxic sesterterpenes, 6-epi-ophiobolin G and 6-epi-ophiobolin N, from marine derived fungus Emericella variecolor GF10 | H Wei, T Itoh, M Kinoshita, Y Nakai, M Kurotaki. 5 July 2004. Tetrahedron. Volume 60, Issue 28,,: Pages 6015-6019.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ophiobolin C, 1 mg | sc-202268 | 1 mg | $300.00 |
