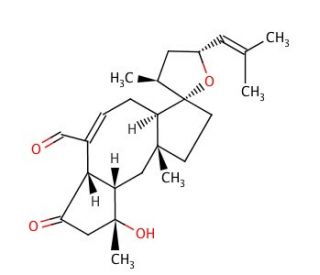
Ophiobolin A (CAS 4611-05-6)
See product citations (7)
QUICK LINKS
Ophiobolin A is a sesterterpenoid compound that is widely studied in the field of plant pathology and mycology due to its role as a phytotoxin produced by pathogenic fungi. It is a subject of research for its ability to induce programmed cell death in plants, which is relevant to understanding plant-fungal interactions and the defense mechanisms of plants. Researchers also utilize Ophiobolin A to investigate its effects on calcium signaling pathways and its potential to disrupt cytoskeletal organization within cells. In the context of natural products chemistry, Ophiobolin A is explored for its complex molecular structure and the biosynthetic pathways leading to its production. Studies involving this compound contribute to the broader knowledge of secondary metabolites produced by fungi and their ecological roles, as well as the potential exploitation of these compounds in agriculture to manage plant diseases.
Ophiobolin A (CAS 4611-05-6) References
- Ophiobolin A from Bipolaris oryzae perturbs motility and membrane integrities of porcine sperm and induces cell death on mammalian somatic cell lines. | Bencsik, O., et al. 2014. Toxins (Basel). 6: 2857-71. PMID: 25251540
- Low concentrations of the toxin ophiobolin A lead to an arrest of the cell cycle and alter the intracellular partitioning of glutathione between the nuclei and cytoplasm. | Locato, V., et al. 2015. J Exp Bot. 66: 2991-3000. PMID: 25890975
- Anticancer activity of Ophiobolin A, isolated from the endophytic fungus Bipolaris setariae. | Bhatia, DR., et al. 2016. Nat Prod Res. 30: 1455-8. PMID: 26212208
- The effect of acute ophiobolin A treatment on HO-mediated inflammatory processes. | Pósa, A., et al. 2017. Hum Exp Toxicol. 36: 594-602. PMID: 27402683
- Influence of light on the biosynthesis of ophiobolin A by Bipolaris maydis. | Fanelli, F., et al. 2017. Nat Prod Res. 31: 909-917. PMID: 27820961
- Ophiobolin A Induces Autophagy and Activates the Mitochondrial Pathway of Apoptosis in Human Melanoma Cells. | Rodolfo, C., et al. 2016. PLoS One. 11: e0167672. PMID: 27936075
- Ophiobolin A, a sesterpenoid fungal phytotoxin, displays different mechanisms of cell death in mammalian cells depending upon the cancer cell origin. | Morrison, R., et al. 2017. Int J Oncol. 50: 773-786. PMID: 28112374
- High ophiobolin A production in endophytic fungus Bipolaris sp. associated with Datura metel. | Maehara, S., et al. 2020. Nat Prod Res. 34: 2990-2992. PMID: 30990079
- Drophiobiolins A and B, Bioactive Ophiobolan Sestertepenoids Produced by Dreschslera gigantea. | Zatout, R., et al. 2020. J Nat Prod. 83: 3387-3396. PMID: 33074690
- Augmented phytotoxic effect of nanoencapsulated ophiobolin A. | Vurro, M., et al. 2022. Nat Prod Res. 36: 1143-1150. PMID: 33342291
- Polygodial and Ophiobolin A Analogues for Covalent Crosslinking of Anticancer Targets. | Maslivetc, V., et al. 2021. Int J Mol Sci. 22: PMID: 34681916
- A Stereoselective Photoinduced Cycloisomerization Inspired by Ophiobolin A. | Law, JA., et al. 2022. Org Lett. 24: 6499-6504. PMID: 35944279
- Ophiobolin A Covalently Targets Complex IV Leading to Mitochondrial Metabolic Collapse in Cancer Cells. | Gowans, FA., et al. 2023. bioRxiv.. PMID: 36945520
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ophiobolin A, 100 µg | sc-202266 | 100 µg | $44.00 | |||
Ophiobolin A, 1 mg | sc-202266A | 1 mg | $250.00 | |||
Ophiobolin A, 5 mg | sc-202266B | 5 mg | $728.00 |
