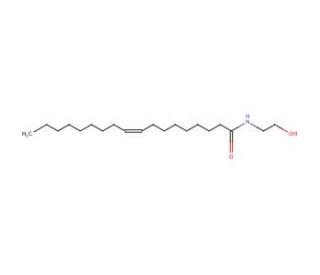
Oleylethanolamide (CAS 111-58-0)
See product citations (1)
QUICK LINKS
Oleylethanolamide is a naturally occurring lipid analogue of the endogenous cannabinoid anandamide (sc-200790). This compound has been noted to be produced in cells and is quickly eliminated by enzymatic hydrolysis. Experiments suggest that Oleylethanolamide causes activation of Neu/ErbB2 receptors and is a selective agonist of GPR55. It is also an endogenous agonist at the GPR119 receptor. Additional studies suggest that this agent can cause Sr/Thr phosphorylation of Glut4 via partial mediation of p38 and JNK kinases. Oleylethanolime has been noted to also regulate the expression of various PPAR-alpha target genes. Oleylethanolamide is an inhibitor of Acid Ceramidase and an activator of PPARalpha and VR1.
Oleylethanolamide (CAS 111-58-0) References
- Quantification of bioactive acylethanolamides in rat plasma by electrospray mass spectrometry. | Giuffrida, A., et al. 2000. Anal Biochem. 280: 87-93. PMID: 10805525
- An anorexic lipid mediator regulated by feeding. | Rodríguez de Fonseca, F., et al. 2001. Nature. 414: 209-12. PMID: 11700558
- Modulation of meal pattern in the rat by the anorexic lipid mediator oleoylethanolamide. | Gaetani, S., et al. 2003. Neuropsychopharmacology. 28: 1311-6. PMID: 12700681
- Oleylethanolamide regulates feeding and body weight through activation of the nuclear receptor PPAR-alpha. | Fu, J., et al. 2003. Nature. 425: 90-3. PMID: 12955147
- Oleylethanolamide impairs glucose tolerance and inhibits insulin-stimulated glucose uptake in rat adipocytes through p38 and JNK MAPK pathways. | González-Yanes, C., et al. 2005. Am J Physiol Endocrinol Metab. 289: E923-9. PMID: 15886223
- Oleylethanolamide activates Ras-Erk pathway and improves myocardial function in doxorubicin-induced heart failure. | Su, HF., et al. 2006. Endocrinology. 147: 827-34. PMID: 16269455
- Role and regulation of acylethanolamides in energy balance: focus on adipocytes and beta-cells. | Matias, I., et al. 2007. Br J Pharmacol. 152: 676-90. PMID: 17704823
- Rapid non-genomic regulation of Ca2+ signals and insulin secretion by PPAR alpha ligands in mouse pancreatic islets of Langerhans. | Ropero, AB., et al. 2009. J Endocrinol. 200: 127-38. PMID: 19017711
- Link between intestinal CD36 ligand binding and satiety induced by a high protein diet in mice. | Naville, D., et al. 2012. PLoS One. 7: e30686. PMID: 22295104
- Effect of High Fat Diets on Body Mass, Oleylethanolamide Plasma Levels and Oxytocin Expression in Growing Rats. | Sospedra, I., et al. 2015. J Food Sci. 80: H1425-31. PMID: 25976631
- Oleoylethanolamine and palmitoylethanolamine modulate intestinal permeability in vitro via TRPV1 and PPARα. | Karwad, MA., et al. 2017. FASEB J. 31: 469-481. PMID: 27623929
- Human Milk and Donkey Milk, Compared to Cow Milk, Reduce Inflammatory Mediators and Modulate Glucose and Lipid Metabolism, Acting on Mitochondrial Function and Oleylethanolamide Levels in Rat Skeletal Muscle. | Trinchese, G., et al. 2018. Front Physiol. 9: 32. PMID: 29472867
- Isotope dilution GC/MS determination of anandamide and other fatty acylethanolamides in rat blood plasma. | Giuffrida, A. and Piomelli, D. 1998. FEBS Lett. 422: 373-6. PMID: 9498819
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Oleylethanolamide, 10 mg | sc-201400 | 10 mg | $90.00 | |||
Oleylethanolamide, 50 mg | sc-201400A | 50 mg | $194.00 |
