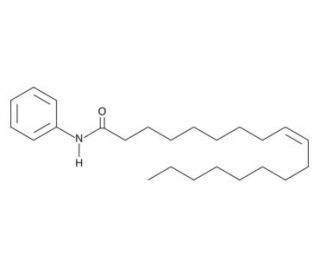
Oleyl Anilide (CAS 5429-85-6)
QUICK LINKS
Oleyl Anilide, bearing the CAS number 5429-85-6, is a chemical compound formed from the condensation of oleyl alcohol, a monounsaturated fatty alcohol, and aniline, a primary aromatic amine. This compound exemplifies a fatty acid amide linkage characterized by the oleyl group, which imparts lipid-like properties, and the anilide functionality, which introduces aromatic character. In research, Oleyl Anilide is primarily used to study surface-active properties in various formulations, particularly in understanding the interactions between lipophilic and aromatic compounds within complex mixtures. Its utility extends to the exploration of molecular self-assembly processes where its amphiphilic nature allows it to stabilize interfaces, forming micelles or other nanostructures in solutions. This ability is crucial in the study of surfactants and emulsifiers in non-medical applications such as in cosmetics, lubricants, and industrial cleaning agents. Additionally, the compound′s structure makes it a candidate for investigating the thermotropic and lyotropic liquid crystalline phases, which are significant in the fields of material science and nanotechnology for creating ordered molecular assemblies. These properties and applications make Oleyl Anilide a valuable subject in the study of organic chemistry and materials engineering, providing insights into the behavior of hybrid organic compounds in various states and conditions.
Oleyl Anilide (CAS 5429-85-6) References
- Epidemiologic evidence for a new class of compounds associated with toxic oil syndrome. | Posada de la Paz, M., et al. 1999. Epidemiology. 10: 130-4. PMID: 10069247
- The Spanish toxic oil syndrome 20 years after its onset: a multidisciplinary review of scientific knowledge. | Gelpí, E., et al. 2002. Environ Health Perspect. 110: 457-64. PMID: 12003748
- Inhibitors of acyl-CoA:cholesterol acyltransferase. 1. Identification and structure-activity relationships of a novel series of fatty acid anilide hypocholesterolemic agents. | Roth, BD., et al. 1992. J Med Chem. 35: 1609-17. PMID: 1578488
- Immunotoxic response of oleic acid anilide and its hydrolysis products in female MRL (+/+) mice. | Cai, P., et al. 2005. J Immunotoxicol. 2: 231-6. PMID: 18958679
- Neurotransmitter changes in cerebrospinal fluid in the Spanish toxic oil syndrome: human clinical findings and experimental results in mice. | del Ser, T., et al. 1986. Neurosci Lett. 67: 135-40. PMID: 2425298
- Role of acyl-CoA: cholesterol acyltransferase in cellular cholesterol metabolism. | Suckling, KE. and Stange, EF. 1985. J Lipid Res. 26: 647-71. PMID: 3897424
- Distribution and excretion of orally administered oleic acid anilide in the rat. | Altmann, HJ. and Grunow, W. 1983. Toxicology. 27: 321-6. PMID: 6623479
- Hydrolysis of oleylanilide in the rat. | Cunningham, VJ., et al. 1983. Arch Toxicol. 54: 157-62. PMID: 6651528
- Decrease of 12-hydroxyeicosatetraenoic acid production in mouse lungs following dietary oleic anilide consumption: implications for the toxic oil syndrome. | Yoshida, SH., et al. 1995. Arch Environ Contam Toxicol. 28: 524-8. PMID: 7755405
- Factors associated with pathogenicity of oils related to the toxic oil syndrome epidemic in Spain. | Posada de la Paz, M., et al. 1994. Epidemiology. 5: 404-9. PMID: 7918809
- Distribution of oleyl-anilide hydrolysing activity in rat and human tissues. | Rodríguez-Martín, A., et al. 1993. Toxicology. 80: 131-9. PMID: 8327996
- Toxic oil syndrome: traceback of the toxic oil and evidence for a point source epidemic. | Posada de la Paz, M., et al. 1996. Food Chem Toxicol. 34: 251-7. PMID: 8621106
- Single step thin-layer chromatographic method for quantitation of enzymatic formation of fatty acid anilides. | Kaphalia, BS. and Ansari, GA. 1998. J Chromatogr B Biomed Sci Appl. 705: 269-75. PMID: 9521563
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Oleyl Anilide, 5 mg | sc-205419 | 5 mg | $20.00 | |||
Oleyl Anilide, 10 mg | sc-205419A | 10 mg | $33.00 |
