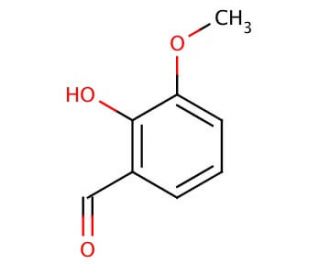
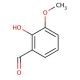
Molecular structure of o-Vanillin, CAS Number: 148-53-8
o-Vanillin (CAS 148-53-8)
Alternate Names:
2-hydroxy-3-methoxybenzaldehyde
CAS Number:
148-53-8
Purity:
≥98%
Molecular Weight:
152.15
Molecular Formula:
C8H8O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
o-Vanillin is an aromatic aldehyde compound widely used in various scientific research applications, particularly in the fields of biochemistry and physiology. For example, it is used in the synthesis of several compounds such as polymers and dyes. o-Vanillin is used in research primarily for its role as a precursor in organic synthesis. Its chemical structure allows for versatile reactions, making it valuable for studying aromatic compound properties and applications.
o-Vanillin (CAS 148-53-8) References
- Free radical scavenging activity of vanillin and o-vanillin using 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical. | Santosh Kumar, S., et al. 2002. Redox Rep. 7: 35-40. PMID: 11981453
- 4-Amino-5-(2-hydroxy-benzyl-idene-amino)benzene-1,2-dicarbonitrile. | Cheng, Y. and Gao, J. 2009. Acta Crystallogr Sect E Struct Rep Online. 65: o963. PMID: 21584006
- Modulating magnetic dynamics of three Dy2 complexes through keto-enol tautomerism of the o-vanillin picolinoylhydrazone ligand. | Guo, YN., et al. 2011. Inorg Chem. 50: 9705-13. PMID: 21902183
- Effects of vanillin and o-vanillin on induction of DNA-repair networks: modulation of mutagenesis in Escherichia coli. | Takahashi, K., et al. 1990. Mutat Res. 230: 127-34. PMID: 2197552
- A vanillin derivative causes mitochondrial dysfunction and triggers oxidative stress in Cryptococcus neoformans. | Kim, JH., et al. 2014. PLoS One. 9: e89122. PMID: 24586538
- Effects of o-vanillin on K⁺ transport of red blood cells from patients with sickle cell disease. | Hannemann, A., et al. 2014. Blood Cells Mol Dis. 53: 21-6. PMID: 24594314
- Vanillin Analogues o-Vanillin and 2,4,6-Trihydroxybenzaldehyde Inhibit NFĸB Activation and Suppress Growth of A375 Human Melanoma. | Marton, A., et al. 2016. Anticancer Res. 36: 5743-5750. PMID: 27793895
- Curcumin and o-Vanillin Exhibit Evidence of Senolytic Activity in Human IVD Cells In Vitro. | Cherif, H., et al. 2019. J Clin Med. 8: PMID: 30934902
- A binuclear CuII/CaII thio-cyanate complex with a Schiff base ligand derived from o-vanillin and ammonia. | Plyuta, N., et al. 2020. Acta Crystallogr E Crystallogr Commun. 76: 423-426. PMID: 32148887
- O-Vanillin Attenuates the TLR2 Mediated Tumor-Promoting Phenotype of Microglia. | Triller, P., et al. 2020. Int J Mol Sci. 21: PMID: 32331440
- Toll-like receptor 2 induced senescence in intervertebral disc cells of patients with back pain can be attenuated by o-vanillin. | Mannarino, M., et al. 2021. Arthritis Res Ther. 23: 117. PMID: 33863359
- o-Vanillin, a promising antifungal agent, inhibits Aspergillus flavus by disrupting the integrity of cell walls and cell membranes. | Li, Q., et al. 2021. Appl Microbiol Biotechnol. 105: 5147-5158. PMID: 34086115
- o-Vanillin Modulates Cell Phenotype and Extracellular Vesicles of Human Mesenchymal Stem Cells and Intervertebral Disc Cells. | Li, L., et al. 2022. Cells. 11: PMID: 36429018
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
o-Vanillin, 10 g | sc-250602 | 10 g | $27.00 | |||
o-Vanillin, 100 g | sc-250602A | 100 g | $91.00 |
