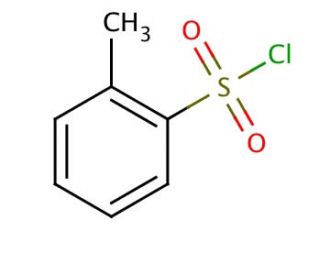
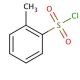
o-Toluenesulfonyl chloride (CAS 133-59-5)
QUICK LINKS
o-Toluenesulfonyl chloride is a chemical compound that functions as a sulfonylating agent in organic synthesis. It is used to introduce the tosyl group into organic molecules, serving as a versatile reagent for the preparation of various tosyl derivatives. The mode of action of o-Toluenesulfonyl chloride involves the substitution of the hydroxyl group in alcohols, phenols, and carboxylic acids, as well as the amino group in amines, to form the corresponding tosylates. This reaction occurs through the displacement of the chloride ion by the nucleophile, resulting in the formation of the tosylated product. o-Toluenesulfonyl chloride can act as a mild dehydrating agent in certain reactions, facilitating the formation of carbon-carbon double bonds. Its ability to selectively modify functional groups makes it a in the synthesis of complex organic molecules for and development purposes.
o-Toluenesulfonyl chloride (CAS 133-59-5) References
- Indolizine 1-sulfonates as potent inhibitors of 15-lipoxygenase from soybeans. | Teklu, S., et al. 2005. Bioorg Med Chem. 13: 3127-39. PMID: 15809148
- Synthesis of optically active oxazoline derivatives via catalytic asymmetric desymmetrization of 1,3-diols. | Tsuda, Y., et al. 2012. Chemistry. 18: 2481-3. PMID: 22298351
- Benzo- and pyrido-1,4-oxazepin-5-ones and -thiones: synthesis and structure-activity relationships of a new series of H1 antihistamines. | Cale, AD., et al. 1989. J Med Chem. 32: 2178-99. PMID: 2570152
- Discovery and SAR of novel pyrazolo[1,5-a]pyrimidines as inhibitors of CDK9. | Phillipson, LJ., et al. 2015. Bioorg Med Chem. 23: 6280-96. PMID: 26349627
- Glutathione Responsive β-Cyclodextrin Conjugated S-Nitrothiols as a Carrier for Intracellular Delivery of Nitric Oxide. | Jia, F., et al. 2019. Bioconjug Chem. 30: 583-591. PMID: 30678457
- Synthesis and Structure-Activity Relationships of Arylsulfonamides as AIMP2-DX2 Inhibitors for the Development of a Novel Anticancer Therapy. | Sivaraman, A., et al. 2020. J Med Chem. 63: 5139-5158. PMID: 32315177
- Discovery of sulfonyl hydrazone derivative as a new selective PDE4A and PDE4D inhibitor by lead-optimization approach on the prototype LASSBio-448: In vitro and in vivo preclinical studies. | Nunes, IKDC., et al. 2020. Eur J Med Chem. 204: 112492. PMID: 32717478
- Motion at the active site of [(4-fluorophenyl)sulfonyl]chymotrypsin. | Ando, ME., et al. 1986. Biochemistry. 25: 4772-8. PMID: 3768312
- Solvation Effects in Hydrolysis of 2-Methylbenzenesulfonyl Halides in Binary Solvents H2O-i-PrOH, H2O-t-BuOH, and H2O-1,4-Dioxane with Low Concentration of Organic Component | , et al. (2004). Russian Journal of General Chemistry. volume 74,: pages 1057–1063.
- Rearrangements of sulfones to sulfinic acids via carbanion intermediates | William E. Truce and William W. Brand. 1970. J. Org. Chem., 35, 6,: 1828–1833.
- Base-promoted rearrangement of arenesulfonamides of N-substituted anilines to N-substituted 2-aminodiaryl sulfones | Sheldon J. Shafer and W. D. Closson. 1975,. J. Org. Chem. 40, 7,: 889–892.
- An approach to C–N activation: coupling of arenesulfonyl hydrazides and arenesulfonyl chlorides with tert-amines via a metal-, oxidant- and halogen-free anodic oxidation†‡ | M. Sheykhan, ORCID logo *a S. Khani,a M. Abbasnia,*bc S. Shaabanzadeha and M. Joafshana. 2017,. Green Chem.,. 19,: 5940-5948.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
o-Toluenesulfonyl chloride, 1 g | sc-228862 | 1 g | $24.00 |
