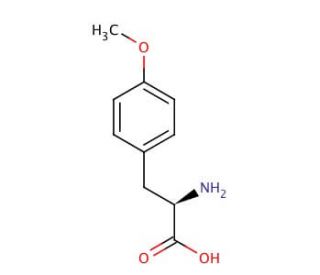
O-Methyl-L-tyrosine (CAS 6230-11-1)
QUICK LINKS
O-Methyl-L-tyrosine (OMT) is a valuable amino acid derivative that has been a mainstay in scientific research for many years. Derived from tyrosine, an indispensable amino acid, O-Methyl-L-tyrosine offers distinctive properties that make it a sought-after compound in various laboratory experiments. The scientific research applications of O-Methyl-L-tyrosine are extensive. It has proven invaluable in drug development studies, where its use helps evaluate the pharmacokinetics, pharmacodynamics, and safety of potential medications. Furthermore, O-Methyl-L-tyrosine has found its place in biochemistry studies, enabling investigations into the intricate structure and function of enzymes. Its utility extends to the study of drug metabolism and transport, as well as the examination of the effects of environmental toxins on the human body. In terms of its mode of action, O-Methyl-L-tyrosine is thought to act as an inhibitor of specific enzymes, such as monoamine oxidase (MAO) and catechol-O-methyltransferase (COMT). Additionally, it is believed to act as an agonist of particular receptors, such as the serotonin receptor.
O-Methyl-L-tyrosine (CAS 6230-11-1) References
- Turning a Substrate Peptide into a Potent Inhibitor for the Histone Methyltransferase SETD8. | Judge, RA., et al. 2016. ACS Med Chem Lett. 7: 1102-1106. PMID: 27994746
- A Robust and Quantitative Reporter System To Evaluate Noncanonical Amino Acid Incorporation in Yeast. | Stieglitz, JT., et al. 2018. ACS Synth Biol. 7: 2256-2269. PMID: 30139255
- Aminoacyl-tRNA Synthetases and tRNAs for an Expanded Genetic Code: What Makes them Orthogonal? | Melnikov, SV. and Söll, D. 2019. Int J Mol Sci. 20: PMID: 31010123
- Biocatalytic Carbon-Hydrogen and Carbon-Fluorine Bond Cleavage through Hydroxylation Promoted by a Histidyl-Ligated Heme Enzyme. | Wang, Y., et al. 2019. ACS Catal. 9: 4764-4776. PMID: 31355048
- Rapid discovery and evolution of orthogonal aminoacyl-tRNA synthetase-tRNA pairs. | Cervettini, D., et al. 2020. Nat Biotechnol. 38: 989-999. PMID: 32284585
- Expanding the Genetic Code for Neuronal Studies. | Nikić-Spiegel, I. 2020. Chembiochem. 21: 3169-3179. PMID: 32531101
- Titrating bacterial growth and chemical biosynthesis for efficient N-acetylglucosamine and N-acetylneuraminic acid bioproduction. | Tian, R., et al. 2020. Nat Commun. 11: 5078. PMID: 33033266
- Metabolic and Lipidomic Assessment of Kidney Cells Exposed to Nephrotoxic Vancomycin Dosages. | Lagies, S., et al. 2021. Int J Mol Sci. 22: PMID: 34576273
- Broadening the Toolkit for Quantitatively Evaluating Noncanonical Amino Acid Incorporation in Yeast. | Stieglitz, JT., et al. 2021. ACS Synth Biol. 10: 3094-3104. PMID: 34730946
- Efficient Unnatural Protein Production by Pyrrolysyl-tRNA Synthetase With Genetically Fused Solubility Tags. | Koch, NG., et al. 2021. Front Bioeng Biotechnol. 9: 807438. PMID: 35284428
- A Chemical Biology Primer for NMR Spectroscopists. | Clark, ET., et al. 2022. J Magn Reson Open. 10-11: PMID: 35494416
- Harnessing the Substrate Promiscuity of Dioxygenase AsqJ and Developing Efficient Chemoenzymatic Synthesis for Quinolones. | Tang, H., et al. 2021. ACS Catal. 11: 7186-7192. PMID: 35721870
- Accessing Chiral Pyrrolodiketopiperazines under Organocatalytic Conditions. | Duñabeitia, E., et al. 2023. Org Lett. 25: 125-129. PMID: 36579971
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
O-Methyl-L-tyrosine, 1 g | sc-219444 | 1 g | $292.00 |
