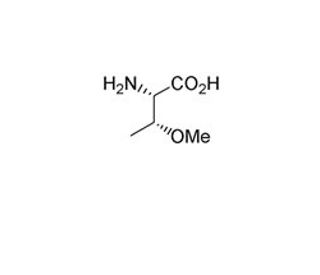
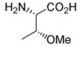
Molecular structure of O-Methyl-L-threonine, CAS Number: 4144-02-9
O-Methyl-L-threonine (CAS 4144-02-9)
Alternate Names:
L-Thr(Me)-OH; (S)-2-Amino-3-methoxybutanoic acid
CAS Number:
4144-02-9
Purity:
≥99%
Molecular Weight:
133.15
Molecular Formula:
C5H11NO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
O-Methyl-L-threonine is a derivative of L-threonine, possessing similar structural characteristics. This compound has been found to effectively suppress the synthesis of fatty acids in resistant mutants, particularly by modifying the amino acid backbone with the addition of a hydroxy group. Additionally, O-Methyl-L-threonine serves as a precursor for the synthesis of nicotinic acetylcholine. It can also be used as photoresists.
O-Methyl-L-threonine (CAS 4144-02-9) References
- Application of capillary electrophoresis with laser-induced fluorescence detection to the determination of biogenic amines and amino acids in brain microdialysate and homogenate samples. | Chen, Z., et al. 2001. J Chromatogr A. 914: 293-8. PMID: 11358224
- Interaction between the synthesis of alpha and beta globin. | Garrick, LM., et al. 1975. Eur J Biochem. 58: 339-50. PMID: 1183442
- [Dependence of the composition of the avermectin complex of Streptomyces avermitilis on glucose content of the growth medium]. | Mironov, VA., et al. 2003. Prikl Biokhim Mikrobiol. 39: 208-12. PMID: 12722656
- Tubulin inhibitors. Synthesis and biological activity of HTI-286 analogs with B-segment heterosubstituents. | Niu, C., et al. 2004. Bioorg Med Chem Lett. 14: 4329-32. PMID: 15261296
- MEKC-LIF of gamma-amino butyric acid in microdialysate: systematic optimization of the separation conditions by factorial analysis. | Ehlen, JC., et al. 2005. J Neurosci Methods. 147: 36-47. PMID: 15979724
- Off-line concomitant release of dopamine and glutamate involvement in taste memory consolidation. | Guzmán-Ramos, K., et al. 2010. J Neurochem. 114: 226-36. PMID: 20412388
- Regio- and Enantioselective Synthesis of Trifluoromethyl-Substituted Homoallylic α-Tertiary NH2 -Amines by Reactions Facilitated by a Threonine-Based Boron-Containing Catalyst. | Fager, DC., et al. 2020. Angew Chem Int Ed Engl. 59: 11448-11455. PMID: 32219997
- Biosynthesis, synthetic studies, and biological activities of the jadomycin alkaloids and related analogues. | de Koning, CB., et al. 2020. Alkaloids Chem Biol. 84: 125-199. PMID: 32416952
- Regulation of isoleucine-valine biosynthesis in Saccharomyces cerevisiae. | Holmberg, S. and Petersen, JG. 1988. Curr Genet. 13: 207-17. PMID: 3289762
- Dipeptides of O-methyl-L-threonine as potential antimalarials. | Gershon, H. and Krull, IS. 1979. J Med Chem. 22: 877-9. PMID: 376849
- On the active site topography of isoleucyl transfer ribonucleic acid synthetase of Escherichia coli B. | Holler, E., et al. 1973. Biochemistry. 12: 1150-9. PMID: 4347457
- Homoserine kinase from Escherichia coli K-12: properties, inhibition by L-threonine, and regulation of biosynthesis. | Théze, J., et al. 1974. J Bacteriol. 118: 577-81. PMID: 4364023
- Enzymes of the isoleucine-valine pathway in Acinetobacter. | Twarog, R. 1972. J Bacteriol. 111: 37-46. PMID: 4669215
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
O-Methyl-L-threonine, 250 mg | sc-295956 | 250 mg | $112.00 | |||
O-Methyl-L-threonine, 1 g | sc-295956A | 1 g | $199.00 |
