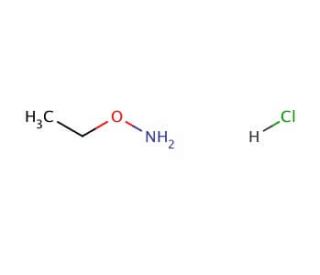
O-Ethylhydroxylamine hydrochloride (CAS 3332-29-4)
QUICK LINKS
O-Ethylhydroxylamine hydrochloride is a chemical reagent that is often employed in synthetic organic chemistry. It is used for the introduction of the ethylhydroxylamino group into organic compounds, serving as a nucleophile in various chemical transformations. One common application is in the conversion of ketones and aldehydes to their corresponding oximes, which can be useful intermediates in the synthesis of other organic molecules. Furthermore, this compound is used in the preparation of nitrones, which are key intermediates in cycloaddition reactions such as the 1,3-dipolar cycloaddition. Researchers also utilize this reagent for the deprotection of certain protecting groups and in the reduction of azides to amines. Its role in these chemical processes is critical for the synthesis of a diverse range of organic compounds, including agrochemicals and materials science precursors.
O-Ethylhydroxylamine hydrochloride (CAS 3332-29-4) References
- Straightforward asymmetric entry to highly functionalized 3-substituted 3-hydroxy-beta-lactams via Baylis-Hillman or bromoallylation reactions. | Alcaide, B., et al. 2004. J Org Chem. 69: 826-31. PMID: 14750811
- Suppression of metabolic defects of yeast isocitrate dehydrogenase and aconitase mutants by loss of citrate synthase. | Lin, AP., et al. 2008. Arch Biochem Biophys. 474: 205-12. PMID: 18359281
- Synthesis and biological evaluation of O-methyl and O-ethyl NSAID hydroxamic acids. | Rajić, Z., et al. 2009. J Enzyme Inhib Med Chem. 24: 1179-87. PMID: 19772490
- Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. | Ito, S., et al. 2011. Science. 333: 1300-3. PMID: 21778364
- Evaluation of different derivatisation approaches for gas chromatographic-mass spectrometric analysis of carbohydrates in complex matrices of biological and synthetic origin. | Becker, M., et al. 2013. J Chromatogr A. 1281: 115-26. PMID: 23399001
- Ethoximation-silylation approach for mono- and disaccharide analysis and characterization of their identification parameters by GC/MS. | Becker, M., et al. 2013. Talanta. 115: 642-51. PMID: 24054643
- Improved quantification of monosaccharides in complex lignocellulosic biomass matrices: A gas chromatography-mass spectrometry based approach. | Zweckmair, T., et al. 2017. Carbohydr Res. 446-447: 7-12. PMID: 28460247
- Effects of glaucocalyxin A on human liver cancer cells as revealed by GC/MS- and LC/MS-based metabolic profiling. | Liu, Y., et al. 2018. Anal Bioanal Chem. 410: 3325-3335. PMID: 29651531
- Usefulness of zebrafish in evaluating drug-induced teratogenicity in cardiovascular system. | Watanabe, R., et al. 2019. Drug Chem Toxicol. 42: 649-656. PMID: 30025483
- Development of an advanced derivatization protocol for the unambiguous identification of monosaccharides in complex mixtures by gas and liquid chromatography. | Haas, M., et al. 2018. J Chromatogr A. 1568: 160-167. PMID: 30122163
- Mechanisms non-enzymatic browning in orange juice during storage. | Paravisini, L. and Peterson, DG. 2019. Food Chem. 289: 320-327. PMID: 30955619
- The intracellular metabolome of starving cells. | Durand, S., et al. 2021. Methods Cell Biol. 164: 137-156. PMID: 34225912
- Simultaneous quantitative analysis of seven steroid hormones in human saliva: A novel method based on O-ethylhydroxylamine hydrochloride as derivatization reagent. | Xu, B., et al. 2022. Rapid Commun Mass Spectrom. 36: e9242. PMID: 34913217
- Effect of oximation reagents on gas chromatographic separation of eight different kinds of mono- and di-saccharides. | Islam, MA., et al. 2022. Food Chem. 386: 132797. PMID: 35344725
- NMR of all-carbon-13 sugars: an application in development of an analytical method for a novel natural sugar, 1,5-anhydrofructose. | Kametani, S., et al. 1996. J Biochem. 119: 180-5. PMID: 8907194
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
O-Ethylhydroxylamine hydrochloride, 1 g | sc-250588 | 1 g | $36.00 | |||
O-Ethylhydroxylamine hydrochloride, 5 g | sc-250588A | 5 g | $77.00 | |||
O-Ethylhydroxylamine hydrochloride, 25 g | sc-250588B | 25 g | $295.00 | |||
O-Ethylhydroxylamine hydrochloride, 75 g | sc-250588C | 75 g | $832.00 |
