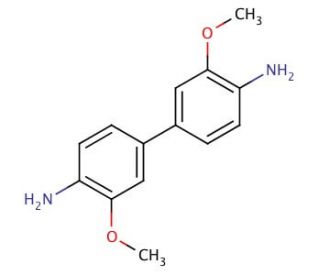
o-Dianisidine (CAS 119-90-4)
QUICK LINKS
o-Dianisidine is an aromatic organic compound extensively employed in diverse scientific and industrial applications. It appears as a colorless solid. Renowned for its versatility, o-Dianisidine serves as a valuable reagent in various realms, including the synthesis of pharmaceuticals, dyes, and other organic compounds. Moreover, it finds substantial use as a biochemical reagent in an extensive range of biological investigations. The applications of o-Dianisidine in scientific research are manifold. It serves as a biochemical reagent, facilitating studies on enzyme kinetics, protein-protein interactions, and signal transduction pathways. Furthermore, it acts as a fluorescent probe for the detection of reactive oxygen species and DNA damage, enabling researchers to explore these important biological phenomena. Additionally, its involvement extends to the synthesis of pharmaceuticals, dyes, and other organic compounds, contributing to the advancement of various fields. The mechanism of action of 3,3′-DMB is contingent upon its specific application. For instance, when utilized as a fluorescent probe, it reacts with reactive oxygen species and DNA damage, leading to the formation of a fluorescent product. As a biochemical reagent, it engages with enzymes and proteins, resulting in the formation of a covalent bond. In the pursuit of biochemical and physiological investigations, 3,3′-DMB has been employed to explore the effects of different compounds on enzyme and protein activity. Additionally, it has been harnessed to scrutinize the impact of various substances on gene and protein expression.
o-Dianisidine (CAS 119-90-4) References
- o-Dianisidine: a new reagent for selective spectrophotometric, flow injection determination of chlorine. | Catalá Icardo, M., et al. 2001. Analyst. 126: 2087-92. PMID: 11763097
- Automated assay of ceruloplasmin by kinetic analysis of o-dianisidine oxidation. | Boyett, JD., et al. 1976. Clin Chim Acta. 69: 233-41. PMID: 1277556
- Use of glucose oxidase, peroxidase, and O-dianisidine in determination of blood and urinary glucose. | HUGGETT, AS. and NIXON, DA. 1957. Lancet. 273: 368-70. PMID: 13464070
- [Detection of o-tolidine and of o-dianisidine and their conversion products in urine and in parts of the organs]. | ENGELBERTZ, P. and BABEL, E. 1954. Zentralbl Arbeitsmed. 4: 179-83. PMID: 14360520
- DNA sensor for o-dianisidine. | Jasnowska, J., et al. 2004. Bioelectrochemistry. 64: 85-90. PMID: 15219251
- Gas diffusion sequential injection system for the spectrophotometric determination of free chlorine with o-dianisidine. | Mesquita, RB. and Rangel, AO. 2005. Talanta. 68: 268-73. PMID: 18970316
- Pyrocatechol as a stabilizing agent for o-tolidine and o-dianisidine: a sensitive new method for HRP neurohistochemistry. | Segade, LA. 1987. J Hirnforsch. 28: 331-40. PMID: 2444642
- Crystal structure of N,N'-dibenzyl-3,3'-di-meth-oxy-benzidine. | Im, H., et al. 2018. Acta Crystallogr E Crystallogr Commun. 74: 271-274. PMID: 29765704
- Tanshinone I, a new EZH2 inhibitor restricts normal and malignant hematopoiesis through upregulation of MMP9 and ABCG2. | Huang, Y., et al. 2021. Theranostics. 11: 6891-6904. PMID: 34093860
- Determination of histaminase (diamine oxidase) activity by o-dianisidine test: interference of ceruloplasmin. | Biegański, T., et al. 1977. Agents Actions. 7: 85-92. PMID: 404858
- Chemical and enzymatic intermediates in the peroxidation of o-dianisidine by horseradish peroxidase. 2. Evidence for a substrate radical--enzyme complex and its reaction with nucleophiles. | Claiborne, A. and Fridovich, I. 1979. Biochemistry. 18: 2329-35. PMID: 444459
- Health hazard alert--benzidine-, o-tolidine-, and o-dianisidine-based dyes DHHS (NIOSH) publication No. 81-106. | . 1981. Am Ind Hyg Assoc J. 42: A36, A38, A40 passim. PMID: 7223641
- An o-dianisidine method of horseradish peroxidase neurohistochemistry. | Takayama, K. and Miura, M. 1981. Jpn J Physiol. 31: 269-72. PMID: 7289229
- Kinetics of oxidation of o-dianisidine by hydrogen peroxide in the presence of antibody complexes of iron(III) coproporphyrin. | Savitsky, AP., et al. 1994. Appl Biochem Biotechnol. 47: 317-27. PMID: 7944346
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
o-Dianisidine, 5 g | sc-215608 | 5 g | $38.00 | |||
o-Dianisidine, 25 g | sc-215608A | 25 g | $189.00 |
