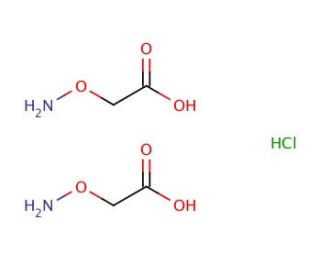
O-(Carboxymethyl)hydroxylamine hemihydrochloride (CAS 2921-14-4)
QUICK LINKS
O-(Carboxymethyl)hydroxylamine hemihydrochloride is a chemical compound that serves a pivotal role in various research fields, particularly in organic and analytical chemistry due to its unique chemical properties and reactivity. Structurally characterized by the presence of a hydroxylamine group attached through an ether linkage to a carboxymethyl moiety, this compound is distinguished by its ability to undergo nucleophilic addition and substitution reactions, making it a versatile reagent for the synthesis of heterocyclic compounds, and for the modification of aldehydes and ketones. Its hemihydrochloride salt form enhances its solubility in aqueous solutions, facilitating its use in a wide range of experimental setups. In analytical applications, it is utilized for the selective detection and quantification of aldehyde and ketone functional groups, leveraging its specificity to form stable adducts, which can be easily identified and measured. This capability is particularly valuable in the analysis of complex organic mixtures and the elucidation of reaction mechanisms, contributing to its prominence as a tool in chemical research endeavors.
O-(Carboxymethyl)hydroxylamine hemihydrochloride (CAS 2921-14-4) References
- The enzymology of alanine aminotransferase (AlaAT) isoforms from Hordeum vulgare and other organisms, and the HvAlaAT crystal structure. | Duff, SM., et al. 2012. Arch Biochem Biophys. 528: 90-101. PMID: 22750542
- Upregulation of cystathionine β-synthetase expression contributes to visceral hyperalgesia induced by heterotypic intermittent stress in rats. | Wang, Y., et al. 2012. PLoS One. 7: e53165. PMID: 23285261
- Neonatal colonic inflammation sensitizes voltage-gated Na(+) channels via upregulation of cystathionine β-synthetase expression in rat primary sensory neurons. | Qu, R., et al. 2013. Am J Physiol Gastrointest Liver Physiol. 304: G763-72. PMID: 23449670
- Sensitization of sodium channels by cystathionine β-synthetase activation in colon sensory neurons in adult rats with neonatal maternal deprivation. | Hu, S., et al. 2013. Exp Neurol. 248: 275-85. PMID: 23834820
- Enhanced synthesis and diminished degradation of hydrogen sulfide in experimental colitis: a site-specific, pro-resolution mechanism. | Flannigan, KL., et al. 2013. PLoS One. 8: e71962. PMID: 23940796
- Upregulation of cystathionine-β-synthetase expression contributes to inflammatory pain in rat temporomandibular joint. | Miao, X., et al. 2014. Mol Pain. 10: 9. PMID: 24490955
- Role of hydrogen sulfide in early blood-brain barrier disruption following transient focal cerebral ischemia. | Jiang, Z., et al. 2015. PLoS One. 10: e0117982. PMID: 25695633
- Sensitization of P2X3 receptors by cystathionine β-synthetase mediates persistent pain hypersensitivity in a rat model of lumbar disc herniation. | Wang, Q., et al. 2015. Mol Pain. 11: 15. PMID: 25885215
- Effect of CCl4 and blocking H2S biosynthesis on oesophageal mucosa rats: model of nonerosive oesophagitis. | Khyrivska, D., et al. 2014. Folia Med Cracov. 54: 79-90. PMID: 25891244
- Efficient Conjugation of Aflatoxin M1 With Bovine Serum Albumin through Aflatoxin M1-(O-carboxymethyl) Oxime and Production of Anti-aflatoxin M1 Antibodies. | Khademi, F., et al. 2015. Jundishapur J Microbiol. 8: e16850. PMID: 26034542
- Upregulation of cystathionine β-synthetase in the arcuate nucleus produces pain hypersensitivity via PKC upregulation and GluN2B phosphorylation in rats with chronic pancreatitis. | Zheng, H., et al. 2016. Sheng Li Xue Bao. 68: 575-584. PMID: 27778022
- Inhibition of cystathionine β-synthetase suppresses sodium channel activities of dorsal root ganglion neurons of rats with lumbar disc herniation. | Yan, J., et al. 2016. Sci Rep. 6: 38188. PMID: 27905525
- Upregulation of Nav1.7 by endogenous hydrogen sulfide contributes to maintenance of neuropathic pain. | Tian, JJ., et al. 2020. Int J Mol Med. 46: 782-794. PMID: 32468069
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
O-(Carboxymethyl)hydroxylamine hemihydrochloride, 250 mg | sc-207410B | 250 mg | $55.00 | |||
O-(Carboxymethyl)hydroxylamine hemihydrochloride, 1 g | sc-207410 | 1 g | $107.00 | |||
O-(Carboxymethyl)hydroxylamine hemihydrochloride, 10 g | sc-207410A | 10 g | $320.00 |
