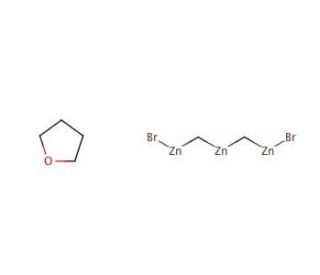
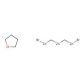
Nysted Reagent (CAS 41114-59-4)
Available in US only.
QUICK LINKS
Nysted reagent is a chemical compound used in synthetic organic chemistry for various applications. Primarily, it serves as a source of the dibromocarbene (:CBr2) species, which is utilized in the addition of dibromomethylene groups to alkenes and alkynes through carbene insertion reactions. These transformations are valuable for the synthesis of dibromocyclopropanes, which are intermediates in the preparation of more complex molecules. The reagent′s utility is further exemplified in the generation of cyclopropane rings, a structural feature present in many natural products and biologically active compounds. Moreover, the Nysted reagent is studied for its role in cyclopropanation reactions under milder conditions compared to other carbene precursors, which can be beneficial when dealing with sensitive substrates. Additionally, researchers are interested in the Nysted reagent′s potential to mediate polymerization processes and the synthesis of polymers with unique properties. Due to its zinc-based composition, the reagent also offers insight into the coordination chemistry of zinc and its role in facilitating organic transformations.
Nysted Reagent (CAS 41114-59-4) References
- Methylenation Reaction of Carbonyl Compounds Using Julia-Kocienski Reagents | Kaori Ando. New Horizons of Process Chemistry.
- Synthesis of stereoisomeric medium-ring alpha,alpha'-dihydroxy cycloalkanones. | Paquette, LA., et al. 2004. J Org Chem. 69: 2454-60. PMID: 15049645
- Asymmetric synthesis of the phytopathogen (+)-fomannosin. | Paquette, LA., et al. 2007. Angew Chem Int Ed Engl. 46: 7817-9. PMID: 17823900
- Exercises in pyrrolidine chemistry: gram scale synthesis of a Pro-Pro dipeptide mimetic with a polyproline type II helix conformation. | Reuter, C., et al. 2011. Chemistry. 17: 12037-44. PMID: 21901773
- An expedient approach to the total synthesis of (+)-5-epi-eudesm-4(15)-ene-1β,6β-diol. | Parthasarathy, G., et al. 2012. Chem Commun (Camb). 48: 11241-3. PMID: 23064601
- Synthesis of indole terpenoid mimics through a functionality-tolerated Eu(fod)3 -catalyzed conjugate addition. | Xiong, X., et al. 2015. Chem Asian J. 10: 869-72. PMID: 25663656
- Recent progress for the synthesis of selected carbocyclic nucleosides. | Bessières, M., et al. 2015. Future Med Chem. 7: 1809-28. PMID: 26416300
- Synthesis of an isomer of lycoplanine A via cascade cyclization to construct the spiro-N,O-acetal moiety. | Gao, W., et al. 2021. Org Biomol Chem. 19: 1748-1751. PMID: 33566055
- Bis(iodozincio)methane - preparation, structure, and reaction | Seijiro Matsubara, Koichiro Oshima, Kiitiro Utimoto. 2001. Journal of Organometallic Chemistry. 617–618: 39-46.
- A one-pot procedure for methylenating carbonyl compounds using the Nysted reagent and titanocene dichloride | Adam Haahr a, Zoran Rankovic b, Richard C. Hartley a. 2011. Tetrahedron Letters. 52: 3020-3022.
- Synthesis of vinylindoles and vinylpyrroles by the Peterson olefination or by use of the Nysted reagent | Wayland E. Noland, Christopher L. Etienne, Nicholas P. Lanzatella. 2011. 48: 381-388.
- Asymmetric synthesis of andavadoic acid via base-catalyzed 5-exo-tet cyclization of a β-hydroperoxy epoxide | Bogdan Barnych a, Bernard Fenet b, Jean-Michel Vatèle a. 2013. Tetrahedron. 69: 334-340.
- Reactivity of cyclohexene epoxides toward intramolecular acid-catalyzed cyclizations for the synthesis of naturally occurring cage architectures | Hajer Abdelkafi, Laurent Evanno 1, Alexandre Deville, Lionel Dubost, Bastien Nay. 2013. Comptes Rendus Chimie. 16: 304-310.
- Domino Methylenation/Hydrogenation of Aldehydes and Ketones by Combining Matsubara's Reagent and Wilkinson's Catalyst | Radhouan Maazaoui, María Pin-Nó, Kevin Gervais, Raoudha Abderrahim, Franck Ferreira, Alejandro Perez-Luna, Fabrice Chemla, Olivier Jackowski. 2016. European Journal of Organic Chemistry. 2016: 5732-5737.
- Diastereoselectivity Switch During Alkene Reductions: Diastereodivergent Syntheses of Molecular Fossils via MHAT or Homogeneous Catalytic Hydrogenation Reactions | Christian R. S. Maior, Paulo C. S. Costa, Carolina B. P. Ligiéro, Lygia S. de Moraes, Gustavo R. Sousa Jr, Prof. Sidney G. Lima, Prof. Pierre M. Esteves, Prof. Paulo C. M. L. Miranda. 2023. Chemistry – A European Journal. 29.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nysted Reagent, 100 g | sc-253205 | 100 g | $151.00 | |||
| US: Only available in the US | ||||||
