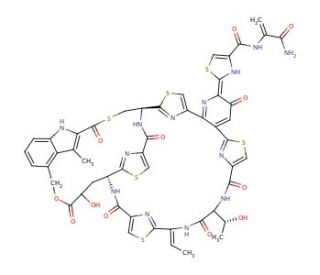
Nosiheptide (CAS 56377-79-8)
QUICK LINKS
Nosiheptide, also known as multhiomycin, is a thiopeptide produced by the bacterium Streptomyces actuosus. It has been extensively studied for its ability to target the 50S subunit of bacterial ribosomes, specifically binding to the 23S rRNA near the peptidyl transferase center. This interaction effectively disrupts the ribosomal function by inhibiting the formation of peptide bonds between amino acids during protein synthesis. This mechanism has made nosiheptide a critical tool for researchers exploring bacterial protein synthesis and ribosome structure. It provides a molecular probe to explain the intricacies of translation inhibition and ribosome dynamics. Structural studies, employing advanced techniques such as X-ray crystallography and cryo-electron microscopy, have benefited from the use of nosiheptide to better understand ribosomal interactions and stability. Additionally, the impact of nosiheptide extends into the field of ribosomal engineering and synthetic biology. Insights gained from its interaction with the ribosome are leveraged to design modified ribosomes with new capabilities, enhancing biotechnological applications.
Nosiheptide (CAS 56377-79-8) References
- Transcriptional organization and regulation of the nosiheptide resistance gene in Streptomyces actuosus. | Li, Y., et al. 1991. J Ind Microbiol. 8: 1-12. PMID: 1367329
- Opportunities and challenges from current investigations into the biosynthetic logic of nosiheptide-represented thiopeptide antibiotics. | Wang, S., et al. 2013. Curr Opin Chem Biol. 17: 626-34. PMID: 23838388
- The importance of start codon of nosM in nosiheptide production. | Jiang, L., et al. 2015. Chin J Nat Med. 13: 854-860. PMID: 26614460
- Enhancing nosiheptide production in Streptomyces actuosus by heterologous expression of haemoprotein from Sinorhizobium meliloti. | Mo, Q., et al. 2016. Lett Appl Microbiol. 62: 480-7. PMID: 27096926
- Total Synthesis of Nosiheptide. | Wojtas, KP., et al. 2016. Angew Chem Int Ed Engl. 55: 9772-6. PMID: 27345011
- Mutagenesis of NosM Leader Peptide Reveals Important Elements in Nosiheptide Biosynthesis. | Jin, L., et al. 2017. Appl Environ Microbiol. 83: PMID: 27913416
- Nosiheptide analogues as potential antibacterial agents via dehydroalanine region modifications: Semi-synthesis, antimicrobial activity and molecular docking study. | Fan, Y., et al. 2021. Bioorg Med Chem. 31: 115970. PMID: 33422909
- Direct determination of nosiheptide residue in animal tissues by liquid chromatography-tandem mass spectrometry. | Shen, J., et al. 2022. J Chromatogr B Analyt Technol Biomed Life Sci. 1193: 123167. PMID: 35196626
- Mutasynthesis Generates Antibacterial Benzothiophenic-Containing Nosiheptide Analogues. | Mu, N., et al. 2022. J Nat Prod. 85: 2274-2281. PMID: 36122372
- Nosiheptide Harbors Potent In Vitro and Intracellular Inhbitory Activities against Mycobacterium tuberculosis. | Yu, X., et al. 2022. Microbiol Spectr. 10: e0144422. PMID: 36222690
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nosiheptide, 1 mg | sc-397320 | 1 mg | $290.00 |
